EZH2, via an association with KDM2B, modulates osteogenic differentiation of root apical papillary stem cells
- PMID: 40308879
- PMCID: PMC12038457
- DOI: 10.4252/wjsc.v17.i4.103482
EZH2, via an association with KDM2B, modulates osteogenic differentiation of root apical papillary stem cells
Abstract
Background: Stem cells from apical papilla (SCAPs) represent promising candidates for bone regenerative therapies due to their osteogenic potential. However, enhancing their differentiation capacity remains a critical challenge. Enhancer of zeste homolog 2 (EZH2), a histone H3 lysine 27 methyltransferase, regulates osteogenesis through epigenetic mechanisms, but its role in SCAPs remains unclear. We hypothesized that EZH2 modulates SCAP osteogenic differentiation via interaction with lysine demethylase 2B (KDM2B), offering a target for therapeutic intervention.
Aim: To investigate the functional role and molecular mechanism of EZH2 in SCAP osteogenic differentiation.
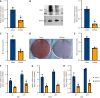
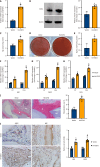
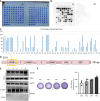
Methods: SCAPs were isolated from healthy human third molars (n = 6 donors). Osteogenic differentiation was assessed via Alizarin red staining and alkaline phosphatase assays. EZH2 overexpression/knockdown models were established using lentiviral vectors. Protein interactions were analyzed by co-immunoprecipitation, transcriptomic changes via microarray (Affymetrix platform), and chromatin binding by chromatin immunoprecipitation-quantitative polymerase chain reaction. In vivo bone formation was evaluated in immunodeficient mice (n = 8/group) transplanted with SCAPs-hydroxyapatite scaffolds. Data were analyzed using Student's t-test and ANOVA.
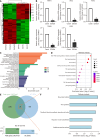
Results: EZH2 overexpression increased osteogenic markers and mineralized nodule formation. In vivo, EZH2-overexpressing SCAPs generated 10% more bone/dentin-like tissue. Co-immunoprecipitation confirmed EZH2-KDM2B interaction, and peptide-mediated disruption of this binding enhanced osteogenesis. Transcriptome analysis identified 1648 differentially expressed genes (971 upregulated; 677 downregulated), with pathway enrichment in Wnt/β-catenin signaling.
Conclusion: EZH2 promotes SCAP osteogenesis via antagonistic interaction with KDM2B, and targeted disruption of this axis offers a translatable strategy for bone regeneration.
Keywords: Apical papillary stem cell; Bioactive peptides; Bone tissue engineering; Enhancer of zeste homolog 2; Osteogenic.
©The Author(s) 2025. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Figures

References
-
- Murab S, Herold S, Hawk T, Snyder A, Espinal E, Whitlock P. Advances in additive manufacturing of polycaprolactone based scaffolds for bone regeneration. J Mater Chem B. 2023;11:7250–7279. - PubMed
-
- Niknam Z, Hosseinzadeh F, Shams F, Fath-Bayati L, Nuoroozi G, Mohammadi Amirabad L, Mohebichamkhorami F, Khakpour Naeimi S, Ghafouri-Fard S, Zali H, Tayebi L, Rasmi Y. Recent advances and challenges in graphene-based nanocomposite scaffolds for tissue engineering application. J Biomed Mater Res A. 2022;110:1695–1721. - PubMed
-
- Fairag R, Rosenzweig DH, Ramirez-Garcialuna JL, Weber MH, Haglund L. Three-Dimensional Printed Polylactic Acid Scaffolds Promote Bone-like Matrix Deposition in Vitro. ACS Appl Mater Interfaces. 2019;11:15306–15315. - PubMed
-
- Eskandani M, Derakhshankhah H, Jahanban-Esfahlan R, Jaymand M. Biomimetic alginate-based electroconductive nanofibrous scaffolds for bone tissue engineering application. Int J Biol Macromol. 2023;249:125991. - PubMed
-
- Yuan B, Wang Z, Zhao Y, Tang Y, Zhou S, Sun Y, Chen X. In Vitro and In Vivo Study of a Novel Nanoscale Demineralized Bone Matrix Coated PCL/β-TCP Scaffold for Bone Regeneration. Macromol Biosci. 2021;21:e2000336. - PubMed
LinkOut - more resources
Full Text Sources

