Physiological magnetic field strengths help magnetotactic bacteria navigate in simulated sediments
- PMID: 40310671
- PMCID: PMC12045620
- DOI: 10.7554/eLife.98001
Physiological magnetic field strengths help magnetotactic bacteria navigate in simulated sediments
Abstract
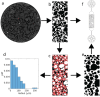
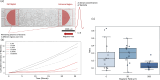
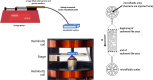
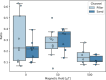
Bacterial motility is typically studied in bulk solution, while their natural habitats often are complex environments. Here, we produced microfluidic channels that contained sediment-mimicking obstacles to study swimming of magnetotactic bacteria in a near-realistic environment. Magnetotactic bacteria are microorganisms that form chains of nanomagnets and that orient in Earth's magnetic field. The obstacles were produced based on micro-computer tomography reconstructions of bacteria-rich sediment samples. We characterized the swimming of the cells through these channels and found that swimming throughput was highest for physiological magnetic fields. This observation was confirmed by extensive computer simulations using an active Brownian particle model. The simulations indicate that swimming at strong fields is impeded by the trapping of bacteria in 'corners' that require transient swimming against the magnetic field for escape. At weak fields, the direction of swimming is almost random, making the process inefficient as well. We confirmed the trapping effect in our experiments and showed that lowering the field strength allows the bacteria to escape. We hypothesize that over the course of evolution, magnetotactic bacteria have thus evolved to produce magnetic properties that are adapted to the geomagnetic field in order to balance movement and orientation in such crowded environments.
Keywords: Magnetospirillum gryphiswaldense; active particles; magnetotaxis; motility; physics of living systems; simulations.
© 2024, Codutti, Charsooghi, Marx et al.
Conflict of interest statement
AC, MC, KM, EC, OM, PZ, VT, TR, DF, SK No competing interests declared
Figures





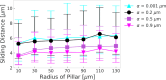
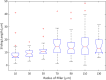



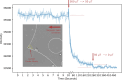
Update of
- doi: 10.1101/2023.12.08.570788
- doi: 10.7554/eLife.98001.1
- doi: 10.7554/eLife.98001.2
References
-
- Bechinger C, Di Leonardo R, Löwen H, Reichhardt C, Volpe G, Volpe G. Active particles in complex and crowded environments. Reviews of Modern Physics. 2016;88:045006. doi: 10.1103/RevModPhys.88.045006. - DOI
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

