Engineering archaeal membrane-spanning lipid GDGT biosynthesis in bacteria: Implications for early life membrane transformations
- PMID: 40313982
- PMCID: PMC12042123
- DOI: 10.1002/mlf2.70001
Engineering archaeal membrane-spanning lipid GDGT biosynthesis in bacteria: Implications for early life membrane transformations
Abstract
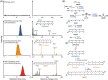
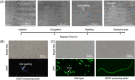
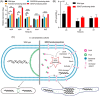
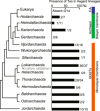
Eukaryotes are hypothesized to be archaeal-bacterial chimeras. Given the different chemical structures of membrane phospholipids in archaea and bacteria, transformations of membranes during eukaryogenesis that led to the bacterial-type membranes of eukaryotic cells remain a major conundrum. One of the possible intermediates of eukaryogenesis could involve an archaeal-bacterial hybrid membrane. So far, organisms with hybrid membranes have not been discovered, and experimentation on such membranes has been limited. To generate mixed membranes, we reconstructed the archaeal membrane lipid biosynthesis pathway in Escherichia coli, creating three strains that individually produced archaeal lipids ranging from simple, such as DGGGOH (digeranylgeranylglycerol) and archaeol, to complex, such as GDGT (glycerol dialkyl glycerol tetraether). The physiological responses became more pronounced as the hybrid membrane incorporated more complex archaeal membrane lipids. In particular, biosynthesis of GDGT induced a pronounced SOS response, accompanied by cellular filamentation, explosive cell lysis, and ATP accumulation. Thus, bacteria seem to be able to incorporate simple archaeal membrane lipids, such as DGGGOH and archaeol, without major fitness costs, compatible with the involvement of hybrid membranes at the early stages of cell evolution and in eukaryogenesis. By contrast, the acquisition of more complex, structurally diverse membrane lipids, such as GDGT, appears to be strongly deleterious to bacteria, suggesting that this type of lipid is an archaeal innovation.
Keywords: SOS response; archaeal lipid GDGT; cellular filamentation; eukaryogenesis; hybrid membrane.
© 2025 The Author(s). mLife published by John Wiley & Sons Australia, Ltd on behalf of Institute of Microbiology, Chinese Academy of Sciences.
Conflict of interest statement
The authors declare no conflict of interests.
Figures

References
-
- Kates M. Diether and tetraether phospholipids and glycolipids as molecular markers for Archaebacteria (Archaea). In: Biology, Environmental Science, Chemistry. Amsterdam: Elsevier Science; 1997. Vol. 671, p. 35–48.
-
- De Rosa M, Gambacorta A, Nicolaus B. A new type of cell membrane, in thermophilic archaebacteria, based on bipolar ether lipids. J Memb Sci. 1983;16:287–294.
-
- Lai D, Springstead JR, Monbouquette HG. Effect of growth temperature on ether lipid biochemistry in Archaeoglobus fulgidus . Extremophiles. 2008;12:271–278. - PubMed
LinkOut - more resources
Full Text Sources
