Herpes simplex virus type-1 infection and spread in a novel porcine corneal explant model is restricted to the epithelium
- PMID: 40315239
- PMCID: PMC12068712
- DOI: 10.1371/journal.ppat.1013162
Herpes simplex virus type-1 infection and spread in a novel porcine corneal explant model is restricted to the epithelium
Abstract
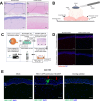
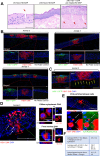
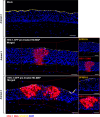
Herpes Keratitis (HK) is a debilitating infection of the cornea that remains the leading cause of infectious blindness in developed countries. Caused primarily by herpes simplex virus type 1 (HSV-1), it is associated with recurrent inflammation, leading to corneal scarring. This study investigated the initial events during acute HSV-1 infection in the cornea by adapting our human anogenital mucosal explant model to a HSV-1 infected porcine corneal explant model. We infected these corneas topically via high-density microarray patches (HD-MAPs) dipped in GFP-labelled HSV-1. Virus infection and spread was detected by both GFP protein and RNAscope, adapted for HSV-1 DNA. The punctures were consistent, usually in the epithelium but some extended into the underlying stroma. However, HSV-1 was restricted to the corneal epithelium, without spread through the anterior limiting membrane (ALM) or Bowman's layer into the stroma nor to the uppermost epithelial layer. This layer expressed SPRR1A similarly to the stratum granulosum of skin which is refractory to HSV-1 infection. In corneas where infected epithelial cells extended to the ALM, SPRR1A was also observed in this layer, suggesting it may contribute to its barrier function. Such studies of HSV-1 infection and spread will help improve therapy for HK and vaccine design to prevent it.
Copyright: © 2025 Arshad et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- Fernando GJP, Hickling J, Jayashi Flores CM, Griffin P, Anderson CD, Skinner SR, et al.. Safety, tolerability, acceptability and immunogenicity of an influenza vaccine delivered to human skin by a novel high-density microprojection array patch (Nanopatch™). Vaccine. 2018;36(26):3779–88. doi: 10.1016/j.vaccine.2018.05.053 - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

