Insulin/PHMB-grafted sodium alginate hydrogels improve infected wound healing by antibacterial-prompted macrophage inflammatory regulation
- PMID: 40319298
- PMCID: PMC12048987
- DOI: 10.1186/s12951-025-03398-8
Insulin/PHMB-grafted sodium alginate hydrogels improve infected wound healing by antibacterial-prompted macrophage inflammatory regulation
Abstract
Background: Non-healing chronic wounds with high susceptibility to infection represent a critical challenge in modern healthcare. While growth factors play a pivotal role in regulating chronic wound repair, their therapeutic efficacy is compromised in infected microenvironments. Current wound dressings inadequately address the dual demands of sustained bioactive molecule delivery and robust antimicrobial activity.
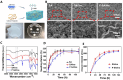
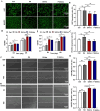
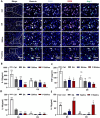
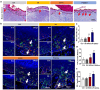
Results: In this study, we developed a sodium alginate hydrogel (termed P-SA/Ins), which incorporated polyhexamethylene biguanide (PHMB) grafting and long-acting glargine insulin loading. P-SA/Ins exhibited the favorable physicochemical performance, biocompatibility and antibacterial efficacy against both Gram-negative and Gram-positive pathogens through inhibition of bacterial proliferation and biofilm formation. Glargine insulin was applied to prolonged insulin delivery. P-SA/Ins treatment attenuated S. aureus induced pro-inflammatory cytokine cascades in macrophages. The evaluation in vivo using a rat model with S. aureus infected wound demonstrated that P-SA/Ins significantly enhanced wound healing and optimized skin barrier through antimicrobial-mediated modulation of macrophage polarization and subsequent inflammatory cytokine profiling.
Conclusions: Our findings demonstrate that P-SA/Ins promotes wound healing and restores epidermal barrier integrity, indicating its potential as a therapeutic dressing for chronic wound healing, particularly in cases with infection risk.
Keywords: Infected wound healing; Inflammation regulation; Insulin; PHMB; Skin barrier.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All animal experiments were approved by the Animal Ethics Committee of Ruijin Hospital Shanghai Jiao Tong University School of Medicine (RJ2023021) and were conducted in compliance with relevant ethical regulations regarding animal testing and research. Patient consent statement: not applicable. Competing interests: The authors declare no competing interests.
Figures




References
-
- Olsson M, Järbrink K, Divakar U, Bajpai R, Upton Z, Schmidtchen A, et al. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair Regen. 2019;27(1):114–25. 10.1111/wrr.12683. - PubMed
-
- ’t Op RC, Walboomers XF, Jansen JA, Wagener F. Design considerations for hydrogel wound dressings: strategic and molecular advances. Tissue Eng Part B Rev. 2020;26(3):230–48. 10.1089/ten.TEB.2019.0281. - PubMed
MeSH terms
Substances
Grants and funding
- 82202442/National Outstanding Youth Science Fund Project of National Natural Science Foundation of China
- 82401034/National Outstanding Youth Science Fund Project of National Natural Science Foundation of China
- 22YF1436700/Shanghai Sailing Program
- YG2025QNB18/Fundamental Research Funds for theCentral Universities
- 82172199/National Natural Science Foundation of China
LinkOut - more resources
Full Text Sources
Medical

