Prognostic Value of Tumor Regression Grade After Chemotherapy Versus Chemoradiotherapy in Patients Undergoing Neoadjuvant Treatment for Locally Advanced Esophageal Adenocarcinoma
- PMID: 40323553
- PMCID: PMC12222383
- DOI: 10.1245/s10434-025-17264-2
Prognostic Value of Tumor Regression Grade After Chemotherapy Versus Chemoradiotherapy in Patients Undergoing Neoadjuvant Treatment for Locally Advanced Esophageal Adenocarcinoma
Abstract
Background: The higher rate of pathological complete response (pCR) after neoadjuvant chemoradiotherapy (NACRT) is an argument to support this treatment. However, previous studies have not demonstrated a survival benefit of NACRT for adenocarcinoma (ADC) compared with neoadjuvant chemotherapy (NACT) and the correlation between pathological tumor response (pTR) and survival is unclear. We aimed to verify whether the prognostic value of pTRis influenced by the type of neoadjuvant treatment performed.
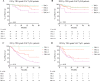
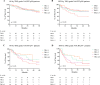
Methods: Patients with ADC who underwent NACT or NACRT and surgery between 2015 and 2020 were included. The correlation between pTR and overall survival (OS) and disease-free survival (DFS) after both treatments was evaluated by using Kaplan-Meier analysis. pTR was assessed by using the Mandard tumor regression grade (TRG).
Results: Overall, 563 patients were included; 278 received NACT, and 285 NACRT. The incidence of pCR was significantly higher after NACRT (24.6% vs. 11.2%, p < 0.0001). The TRG of both node-negative (pN0) and node-positive (pN+) patients significantly correlated with the 5 years OS after NACT (pN0 p = 0.03, pN+ p = 0.01). The same result was not detected in NACRT patients (pN0 p = 0.98, pN+ p = 0.23). The 5-year DFS of the patients with pCR was higher in the NACT group (84% vs. 66.5%, p = 0.05). The proportion of patients showing distant recurrences was significantly higher in the NACRT group (35.4% vs. 23.8%, p = 0.009).
Conclusions: Tumor regression grade was significantly associated with survival after NACT, but not with NACRT. Despite a lower rate of pCR, both OS and, especially, DFS of patients with pCR improved after NACT compared with NACRT.
© 2025. The Author(s).
Conflict of interest statement
Disclosure: Sara Lonardi: Research funding (to Institution) from Amgen, Astellas, Astra Zeneca, Bayer, Bristol-Myers Squibb, Daichii Sankyo, Hutchinson, Incyte, Merck Serono, Mirati, MSD, Pfizer, Roche, Servier, personal honoraria as invited speaker from Amgen, Astra Zeneca, Bristol-Myers Squibb, Incyte, GSK, Lilly, Merck Serono, MSD, Pierre-Fabre, Roche, Servier; Participation in advisory board for Amgen, Astellas, Astra Zeneca, Bayer, Bristol-Myers Squibb, Daiichi-Sankyo, GSK, Incyte, Lilly, Merck Serono, MSD, Servier, Takeda, Rottapharm, Beigene, Fosum, Nimbus Therapeutics. Peter Grimminger: Proctor for Intuitive Surgical, Advisor for Medicaroid Europe, Medtronic, Richard Wolf and JnJ.
Figures

References
-
- Reynolds JV, Preston SR, O’Neill B, et al. Trimodality therapy versus perioperative chemotherapy in the management of locally advanced adenocarcinoma of the oesophagus and oesophagogastric junction (Neo-AEGIS): an open-label, randomised, phase 3 trial. Lancet Gastroenterol Hepatol. 2023;8(11):1015–27. 10.1016/S2468-1253(23)00243-1. - DOI - PMC - PubMed
Publication types
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical

