Nageotte nodules in human dorsal root ganglia reveal neurodegeneration in diabetic peripheral neuropathy
- PMID: 40325011
- PMCID: PMC12052976
- DOI: 10.1038/s41467-025-59538-z
Nageotte nodules in human dorsal root ganglia reveal neurodegeneration in diabetic peripheral neuropathy
Abstract
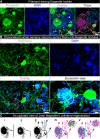
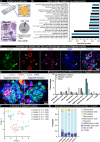
Nageotte nodules, first described in 1922 by Jean Nageotte, are clusters of non-neuronal cells that form after sensory neuron death. Despite their historical recognition, little is known about their molecular identity nor their involvement in neuropathies that involve neuronal loss like diabetic peripheral neuropathy (DPN). In this study, we molecularly characterize Nageotte nodules in dorsal root ganglia recovered from organ donors with DPN. Here we show that Nageotte nodules are abundant in DPN sensory ganglia and account for 25% of all neurons. Peripherin-and Nav1.7-positive dystrophic axons invade Nageotte nodules, forming small neuroma-like structures. Using histology and spatial sequencing, we demonstrate that Nageotte nodules are mainly composed of satellite glia and non-myelinating Schwann cells that express SPP1 and are intertwined with sprouting sensory axons originating from neighboring neurons. Our findings suggest that Nageotte nodules are an integral feature of dorsal root ganglion neurodegeneration, providing potential therapeutic targets for sensory neuron protection and pain management in DPN.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: T.J.P. is a co-founder of 4E Therapeutics. The authors declare no other competing interests related to this work.
Figures



Update of
-
Nageotte nodules in human DRG reveal neurodegeneration in painful diabetic neuropathy.bioRxiv [Preprint]. 2024 Aug 23:2024.08.22.609215. doi: 10.1101/2024.08.22.609215. bioRxiv. 2024. Update in: Nat Commun. 2025 May 5;16(1):4168. doi: 10.1038/s41467-025-59538-z. PMID: 39229145 Free PMC article. Updated. Preprint.
-
Nageotte nodules in human DRG reveal neurodegeneration in painful diabetic neuropathy.Res Sq [Preprint]. 2024 Sep 27:rs.3.rs-5006011. doi: 10.21203/rs.3.rs-5006011/v1. Res Sq. 2024. Update in: Nat Commun. 2025 May 5;16(1):4168. doi: 10.1038/s41467-025-59538-z. PMID: 39399674 Free PMC article. Updated. Preprint.
References
-
- Nageotte, J. L’organisation de la matiere dans ses rapports avec la vie: etudes d’anatomie generale et de morphologie experimentale sur le tissu conjonctif et le nerf. (F. Alcan, 1922).
MeSH terms
Substances
Grants and funding
- R01 NS111929/NS/NINDS NIH HHS/United States
- U19 NS130608/NS/NINDS NIH HHS/United States
- R01NS111929/U.S. Department of Health & Human Services | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
- U19NS130608/U.S. Department of Health & Human Services | NIH | National Institute of Neurological Disorders and Stroke (NINDS)
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

