A murine model of Barth syndrome recapitulates human cardiac and skeletal muscle phenotypes
- PMID: 40326536
- PMCID: PMC12128220
- DOI: 10.1242/dmm.052077
A murine model of Barth syndrome recapitulates human cardiac and skeletal muscle phenotypes
Abstract
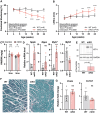
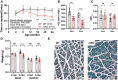
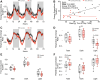
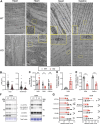
Barth syndrome is a mitochondrial disorder with hallmarks of cardiac and skeletal muscle weakness. It is caused by pathogenic variants in the X-linked gene tafazzin (TAZ), required for cardiolipin remodeling. Previously described germline and conditional Taz knockout models are not ideal for therapeutic development because they lack the combination of robust survival to adulthood, cardiomyopathy and skeletal muscle weakness. We characterized a cardiac and skeletal muscle-specific Taz knockout model (TazmKO) in which Cre recombinase is expressed from the muscle creatine kinase promoter (mCK-Cre). TazmKO mice survived normally. Cardiolipin composition was abnormal in both heart and skeletal muscle. TazmKO had reduced heart function by 2 months of age, and function progressively declined thereafter. Reduced treadmill endurance and diminished peak oxygen consumption were evident by 3 months of age, suggesting reduced skeletal muscle function. Electron microscopy showed abnormalities in mitochondrial structure and distribution. Overall, TazmKO mice display diminished cardiac function and exercise capacity while maintaining normal survival. This model will be useful for studying the effects of TAZ deficiency in striated muscles and for testing potential therapies for Barth syndrome.
Keywords: Barth syndrome; Cardiomyopathy; Skeletal myopathy; Tafazzin.
© 2025. Published by The Company of Biologists.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures


References
-
- Bashir, A., Bohnert, K. L., Reeds, D. N., Peterson, L. R., Bittel, A. J., de Las Fuentes, L., Pacak, C. A., Byrne, B. J. and Cade, W. T. (2017). Impaired cardiac and skeletal muscle bioenergetics in children, adolescents, and young adults with Barth syndrome. Physiol. Rep. 5, e13130. 10.14814/phy2.13130 - DOI - PMC - PubMed
-
- Brüning, J. C., Michael, M. D., Winnay, J. N., Hayashi, T., Hörsch, D., Accili, D., Goodyear, L. J. and Kahn, C. R. (1998). A muscle-specific insulin receptor knockout exhibits features of the metabolic syndrome of NIDDM without altering glucose tolerance. Mol. Cell 2, 559-569. 10.1016/S1097-2765(00)80155-0 - DOI - PubMed
-
- Chatfield, K. C., Sparagna, G. C., Specht, K. S., Whitcomb, L. A., Omar, A. K., Miyamoto, S. D., Wolfe, L. M. and Chicco, A. J. (2022). Long-chain fatty acid oxidation and respiratory complex I deficiencies distinguish Barth Syndrome from idiopathic pediatric cardiomyopathy. J. Inherit. Metab. Dis. 45, 111-124. 10.1002/jimd.12459 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

