Effects of discontinuing different antiresorptive regimens on medication-related osteonecrosis of the jaw in patients undergoing dental procedures: a systematic review and network meta-analysis
- PMID: 40326547
- PMCID: PMC12061011
- DOI: 10.1530/EOR-2024-0133
Effects of discontinuing different antiresorptive regimens on medication-related osteonecrosis of the jaw in patients undergoing dental procedures: a systematic review and network meta-analysis
Abstract
Purpose: Controversy exists on whether a drug holiday is necessary for patients on antiresorptive medication for osteoporosis or bone metastasis and undergoing dental procedures to lower the risk of medication-related osteonecrosis of the jaw (MRONJ). This study evaluated the effects of discontinuing different antiresorptive regimens on MRONJ in these patients.
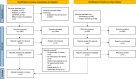
Methods: Publications from PubMed, EMBASE, Cochrane Library and EBSCO Open Dissertations were searched from inception to September 2023 following PRISMA guidelines, and the review was registered in PROSPERO. Eligibility criteria included clinical studies on the effects of continued and discontinued antiresorptive medications for osteoporosis or bone metastasis in patients undergoing dental procedures. The involved antiresorptive agents were oral bisphosphonates (BPs), intravenous (IV) BPs and denosumab (Dmab). Relative risk (RR) with 95% confidence interval (CI) was estimated using a random-effects model.
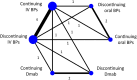
Results: Of the 2,590 records identified, six studies (n = 717) were included. Discontinued use of oral BPs had a lower MRONJ risk than discontinuation of IV BPs (RR = 0.05; 95% CI: 0.00-0.83) and continuation of IV BPs (RR = 0.03; 95% CI: 0.00-0.46). Continuing oral BPs also resulted in a lower MRONJ risk compared to both discontinuation and continuation of IV BPs, with RR = 0.04 (95% CI: 0.00-0.67) and RR = 0.03 (95% CI: 0.00-0.37), respectively. No significant difference was found between continuation and discontinuation of oral BPs, along with other comparisons.
Conclusions: A drug holiday may not be necessary before dental procedures for oral BPs. Temporary discontinuation of IV BPs or Dmab is also unlikely to reduce MRONJ risk compared to continued medication.
Keywords: antiresorptives; bisphosphonates; bone metastasis; denosumab; osteonecrosis of the jaw; osteoporosis.
Conflict of interest statement
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the work reported.
Figures
References
-
- Camacho PM, Petak SM, Binkley N, et al. American Association of Clinical Endocrinologists/American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis-2020 update. Endocr Pract 2020. 26(Supp. 1) 1–46. ( 10.4158/GL-2020-0524SUPPL) - DOI - PubMed
LinkOut - more resources
Full Text Sources

