Mossy fiber expression of αSMA in human hippocampus and its relevance to brain evolution and neuronal development
- PMID: 40328887
- PMCID: PMC12056149
- DOI: 10.1038/s41598-025-00094-3
Mossy fiber expression of αSMA in human hippocampus and its relevance to brain evolution and neuronal development
Abstract
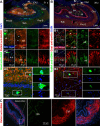
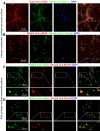
α-Smooth muscle actin (αSMA) is best characterized as the building block of thin filaments in smooth muscle cells. We observed a clear αSMA immunolabeling in adult human hippocampal mossy fibers (MF), prompting us to explore this novel pattern in phylogenic and ontogenic perspectives in the present study. αSMA immunolabeling occurred distinctively at the hippocampal MF terminals in humans from infancy to elderly. Hippocampal MF αSMA immunolabeling was not observed in mice and rats, visible in CA3 in guinea pigs and cats, and prominent in CA3 and dentate hilus in Rhesus monkeys. MF αSMA immunolabeling in human hippocampus emerged and refined from the last gestational trimester to early infancy. A transient overall neuronal labeling of ɑSMA was observed in prenatal human brains. ɑSMA expression was detected in human and rat primary neuronal cultures. The specificity of ɑSMA antibodies was confirmed by ACTA2 small interfering RNA (siRNA) silencing in SH-SY5Y cells. With this validation, we detected a higher αSMA protein level in dentate gyrus lysates relative to other human brain areas. Taken together, αSMA is distinctly expressed in human hippocampal mossy fibers. This pattern is related to hippocampal evolution among mammals and involves a refinement of neuronal αSMA expression during brain development.
Keywords: Actin isoforms; Brain development; Mammalian evolution; Synaptoplasticity; Trisynaptic circuit.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics approval: All human brains were banked with written informed consent obtained prior to post-mortem donation from the donor and/or the next-of kin. The Ethics Committee for Research and Education at Xiangya School of Medicine approved the use of postmortem human materials. Animal brain sections were obtained from previous original studies, with animal use and experimental protocols approved institutionally and documented in corresponding original reports.
Figures





References
-
- Cheever, T. R. & Ervasti, J. M. Actin isoforms in neuronal development and function. Int. Rev. Cell Mol. Biol.301, 157–213 (2013). - PubMed
-
- Dugina, V., Shagieva, G. & Kopnin, P. Biological role of actin isoforms in mammalian cells. Biochem. Mosc.84, 583–592 (2019). - PubMed
-
- Gunning, P. W., Ghoshdastider, U., Whitaker, S., Popp, D. & Robinson, R. C. The evolution of compositionally and functionally distinct actin filaments. J. Cell Sci.128, 2009–2019 (2015). - PubMed
MeSH terms
Substances
Grants and funding
- 82201595/National Natural Science Foundation of China
- 82071223/National Natural Science Foundation of China
- grant #2021ZD0201103/Ministry of Science and Technology of the People's Republic of China
- 2021ZD02018/Ministry of Science and Technology of the People's Republic of China
- 2020SK2122/Science and Technology Program of Hunan Province
LinkOut - more resources
Full Text Sources
Miscellaneous

