Macrophage response to fibrin structure mediated by Tgm2-dependent mitochondrial mechanosensing
- PMID: 40331213
- PMCID: PMC12051126
- DOI: 10.1016/j.bioactmat.2025.04.022
Macrophage response to fibrin structure mediated by Tgm2-dependent mitochondrial mechanosensing
Abstract
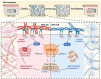
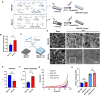
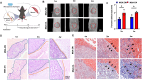
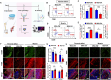
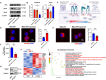
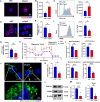
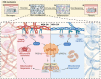
Following an injury at the implantation position, blood-material interactions form a fibrin architecture, which serves as the initial activator of foreign body response (FBR). However, there is limited knowledge regarding how the topography of fibrin architectures regulates macrophage behavior in mitigating FBR. Mechanical cues of the microenvironment have been reported to shape immune cell functions. Here, we investigated macrophage mechanobiology at the organelle level by constructing heterogeneous fibrin networks. Based on findings in vivo, we demonstrated that adhesion-mediated differentiation of mitochondrial function modulated macrophage polarization. The finite activation of integrin signaling upregulated transglutaminase 2 (Tgm2) in a trans-manner, augments PGC1α-mediated mitochondrial biogenesis. Our study highlighted the previously overlooked spatial structures of host proteins adsorbed on material surfaces, advocating for a paradigm shift in material design strategies, from focusing solely on physical properties to considering the modification of host proteins.
Keywords: Biomaterial recognition; Foreign body response; Mechanotransduction; Mitochondrion; Transglutaminase.
© 2025 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


Similar articles
-
Regulating Blood Clot Fibrin Films to Manipulate Biomaterial-Mediated Foreign Body Responses.Research (Wash D C). 2023 Sep 15;6:0225. doi: 10.34133/research.0225. eCollection 2023. Research (Wash D C). 2023. PMID: 37719049 Free PMC article.
-
The extended effect of adsorbed damage-associated molecular patterns and Toll-like receptor 2 signaling on macrophage-material interactions.Front Bioeng Biotechnol. 2022 Aug 26;10:959512. doi: 10.3389/fbioe.2022.959512. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36091432 Free PMC article.
-
Fibrin polymer on the surface of biomaterial implants drives the foreign body reaction.Biomaterials. 2021 Oct;277:121087. doi: 10.1016/j.biomaterials.2021.121087. Epub 2021 Aug 25. Biomaterials. 2021. PMID: 34478933 Free PMC article.
-
Beyond Encapsulation: Exploring Macrophage-Fibroblast Cross Talk in Implant-Induced Fibrosis.Tissue Eng Part B Rev. 2024 Dec;30(6):596-606. doi: 10.1089/ten.TEB.2023.0300. Epub 2024 Mar 27. Tissue Eng Part B Rev. 2024. PMID: 38420650 Review.
-
Macrophages and fibroblasts in foreign body reactions: How mechanical cues drive cell functions?Mater Today Bio. 2023 Aug 29;22:100783. doi: 10.1016/j.mtbio.2023.100783. eCollection 2023 Oct. Mater Today Bio. 2023. PMID: 37701130 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

