A Cross-Sectional Study Demonstrating a High Prevalence of Skin Rash to Diabetes Medical Devices: An Underestimated Problem
- PMID: 40331898
- PMCID: PMC12058707
- DOI: 10.1177/19322968251336261
A Cross-Sectional Study Demonstrating a High Prevalence of Skin Rash to Diabetes Medical Devices: An Underestimated Problem
Abstract
Background: Adverse skin reactions to continuous glucose monitors (CGMs) and devices for continuous subcutaneous insulin infusions (CSIIs) ("diabetes medical devices" (MDs)) are well known. However, epidemiological studies on prevalence and skin rash details are lacking. The objective of this study was to describe the prevalence and details of skin rash to diabetes MDs in adults with type 1 diabetes.
Method: All adult individuals (≥18 years) with type 1 diabetes attending outpatient diabetes clinics at two hospitals in southern Sweden were invited to participate (n = 1943) in a questionnaire study.
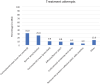
Results: The questionnaire was completed by 667. Of the respondents 95.1% had used CGM and 36.7% had used CSII. Skin rash to the devices was reported by 42.1% of CGM users and 44.9% of CSII users. Skin rash was reported with use of all types of diabetes MDs. For diabetes MDs with ≥50 users, 18.0% to 56.5% of the participants with skin rash had to change the device more often than recommended and 4.0% to 18.0% had to stop using the device due to skin rash. In multivariable analyses, the odds for skin rash to diabetes MDs were higher among younger individuals and individuals with childhood atopic dermatitis. Odds increased with use of higher numbers of devices. Of the participants with skin rash, 13 of the 289 (4.5%) had been investigated for contact allergy.
Conclusions: Skin rash to diabetes MDs is common. The problem is underdiagnosed in clinical practice. With use of diabetes MDs expected to increase, an increasing prevalence of skin rash is to be expected.
Keywords: contact dermatitis; continuous glucose monitoring; insulin infusion systems; medical device; type 1 diabetes.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MB is a member of the expert panel for fragrance safety (
Figures
References
-
- Nathan DM, Cleary PA, Backlund JY, et al. Diabetes control and complications trial/epidemiology of diabetes interventions and complications (DCCT/EDIC) study research group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353(25):2643-2653. doi: 10.1056/NEJMoa052187. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

