Non-Invasive Stratification of Portal Hypertension in Patients With BCR::ABL1-Negative Myeloproliferative Neoplasms
- PMID: 40332039
- PMCID: PMC12057651
- DOI: 10.1111/liv.70098
Non-Invasive Stratification of Portal Hypertension in Patients With BCR::ABL1-Negative Myeloproliferative Neoplasms
Abstract
Background & aims: The course of BCR::ABL1-negative myeloproliferative neoplasms (MPN) is frequently complicated by thromboembolic events in the splanchnic venous system, resulting in portal hypertension (PH). Therefore, the introduction of spleen stiffness measurement (SSM) might improve the diagnosis of PH. The aim of this study was to evaluate the clinical utility of SSM (performed by using the 100 Hz probe) in non-invasive stratification of PH in these patients.
Methods: We performed a retrospective, monocentric, cross-sectional analysis including consecutive patients with BCR::ABL1-negative MPN attending the haematological outpatient clinic at the Medical University of Vienna with available liver stiffness (LSM)/SSM from 10/2023 to 09/2024. LSM/SSM were linked to signs and events of PH.
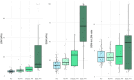
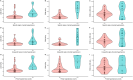
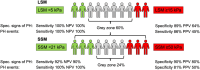
Results: Fifty-five patients were included (mean age 57.9 ± 14.2 years, 69% females, polycythaemia vera as main entity). One fourth of patients had splanchnic vein thrombi. Nineteen patients (34.5%) had specific and 29 patients (52.7%) had unspecific signs of PH. Twelve patients (21.8%) experienced PH events prior to study inclusion. SSM correlated with disease severity (i.e., JAK2 V617F allele frequency). LSM/SSM adequately stratified patients with vs. without PH. While SSM was strongly linked with splenomegaly, it yielded independent information regarding PH on top of splenomegaly. The implementation of sequential LSM (< 5 & ≥ 15 kPa) and SSM (< 21 & ≥ 50 kPa) for ruling in and out PH reduced the grey zone (24%) with adequate sensitivity/specificity.
Conclusions: While SSM is strongly correlated with splenomegaly and disease severity, it is independently associated with PH in patients with BCR::ABL1-negative MPN. Implementation of LSM/SSM might improve patient management.
Keywords: MPN; PSVD; SSM; portal hypertension; spleen stiffness.
© 2025 The Author(s). Liver International published by John Wiley & Sons Ltd.
Conflict of interest statement
Tobias Meischl received speaker honoraria from AstraZeneca, Chiesi, Janssen‐Cilag, Novartis, and sobi; consulting fees from CSL Behring; and travel support from AstraZeneca, BeiGene, CSL Behring, Chiesi, Gilead, Jazz Pharmaceuticals, Janssen‐Cilag, Novartis, and teva‐ratiopharm. Mattias Mandorfer served as a speaker and/or consultant and/or advisory board member for AbbVie, Gilead, Collective Acumen, and W. L. Gore & Associates, Takeda and received travel support from AbbVie, Bristol‐Myers Squibb, and Gilead. Thomas Reiberger served as a speaker and/or consultant and/or advisory board member for AbbVie, Bayer, Boehringer Ingelheim, Gilead, Intercept, MSD, Roche, Siemens, and W. L. Gore & Associates and received grants/research support from AbbVie, Boehringer Ingelheim, Gilead, MSD, Philips, and W. L. Gore & Associates as well as travel support from Boehringer Ingelheim and Gilead. Michael Trauner received speaker fees from BMS, Falk Foundation, Gilead, Intercept, Ipsen, Janssen, Madrigal, MSD, and Roche; he advised for Agomab, AbbVie, Albireo, BiomX, Boehringer Ingelheim, Chemomab, Cymabay, Falk Pharma GmbH, Genfit, Gilead, Hightide, Intercept, Ipsen, Janssen, Mirum, MSD, Novartis, Phenex, Pliant, Rectify, Regulus, Siemens, and Shire. He further received travel support from AbbVie, Falk, Gilead, Intercept, and Janssen and research grants from Albireo, Alnylam, Cymabay, Falk, Gilead, Intercept, MSD, Takeda, and UltraGenyx. He is also a co‐inventor of patents on the medical use of norUDCA filed by the Medical Universities of Graz and Vienna. Bernhard Scheiner received travel support from AbbVie, Ipsen, and Gilead. Georg Semmler received travel support from Amgen. The other authors declare no conflicts of interest.
Figures


References
-
- Spivak J. L., “Myeloproliferative Neoplasms,” New England Journal of Medicine 376, no. 22 (2017): 2168–2181. - PubMed
-
- Smalberg J. H., Arends L. R., Valla D. C., Kiladjian J. J., Janssen H. L. A., and Leebeek F. W. G., “Myeloproliferative Neoplasms in Budd‐Chiari Syndrome and Portal Vein Thrombosis: A Meta‐Analysis,” Blood 120, no. 25 (2012): 4921–4928. - PubMed
-
- Hoekstra J., Bresser E. L., Smalberg J. H., Spaander M. C., Leebeek F. W., and Janssen H. L., “Long‐Term Follow‐Up of Patients With Portal Vein Thrombosis and Myeloproliferative Neoplasms,” Journal of Thrombosis and Haemostasis 9, no. 11 (2011): 2208–2214. - PubMed
-
- Singh S., Muir A. J., Dieterich D. T., and Falck‐Ytter Y. T., “American Gastroenterological Association Institute Technical Review on the Role of Elastography in Chronic Liver Diseases,” Gastroenterology 152, no. 6 (2017): 1544–1577. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

