Enzalutamide and Prostate-Specific Antigen Levels in Metastatic Prostate Cancer: A Secondary Analysis of the ARCHES Randomized Clinical Trial
- PMID: 40332939
- PMCID: PMC12059972
- DOI: 10.1001/jamanetworkopen.2025.8751
Enzalutamide and Prostate-Specific Antigen Levels in Metastatic Prostate Cancer: A Secondary Analysis of the ARCHES Randomized Clinical Trial
Abstract
Importance: In men with metastatic hormone-sensitive prostate cancer (mHSPC), prostate-specific antigen (PSA) decline after treatment has been associated with improved survival. However, the data on PSA decline are limited in men with mHSPC after treatment with enzalutamide plus androgen deprivation therapy (ADT).
Objective: To evaluate the association of enzalutamide plus ADT in men with mHSPC by PSA level at study enrollment in individuals with prior ADT and by degree of PSA reduction with clinical end points.
Design, setting, and participants: This post hoc secondary analysis was performed on data collected from the ARCHES multinational, double-blind, phase 3 randomized clinical trial. In the ARCHES trial, men with mHSPC were randomized between March 21, 2016, and January 12, 2018. Analyses were performed in October 2018 (median follow-up, 14.4 months; IQR, 11.2-17.7 months) and May 2021 (median follow-up, 44.6 months; IQR, 41.3-48.6 months).
Intervention: Patients were randomized 1:1 to enzalutamide (160 mg/d) plus ADT or placebo plus ADT. ADT received for 3 to 6 months before study enrollment was permitted.
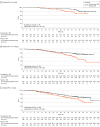
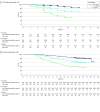
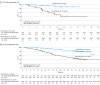
Main outcomes and measures: Radiographic progression-free survival (rPFS) and overall survival (OS) were correlated post hoc with PSA level at enrollment in patients with prior ADT and additionally with PSA decline at 6 months or undetectable PSA (<0.2 ng/mL) during study treatment.
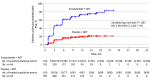
Results: A total of 1150 men (median age, 70 years; range, 46-92 years) were enrolled. Improvement in clinical outcomes such as rPFS was seen with enzalutamide plus ADT in men with PSA levels at enrollment of 0.2 to 4 ng/mL and above 4 ng/mL. Hazard ratios (HRs) for improved rPFS in men with PSA levels up to 0.2 ng/mL, above 0.2 to 4 ng/mL, and above 4 ng/mL were 0.59 (95% CI, 0.27-1.30), 0.32 (95% CI, 0.20-0.50), and 0.44 (95% CI, 0.32-0.62), respectively. Patients treated with enzalutamide plus ADT who achieved undetectable PSA had an 86.0% reduced risk of radiographic disease progression (HR, 0.14 [95% CI, 0.09-0.23]; P < .001) and a 76.0% reduced risk of death (HR, 0.24 [95% CI, 0.17-0.34]; P < .001).
Conclusions and relevance: In this secondary analysis of a randomized clinical trial, patients with mHSPC treated with enzalutamide plus ADT vs placebo plus ADT had improved clinical outcomes regardless of PSA level at enrollment. Undetectable PSA was associated with improved clinical outcomes with enzalutamide treatment. Future studies are required to further characterize the clinical utility of further treatment intensification among men with mHSPC for whom ADT plus enzalutamide fails to achieve undetectable PSA.
Trial registration: ClinicalTrials.gov Identifier: NCT02677896.
Conflict of interest statement
Figures

References
-
- Xtandi highlights of prescribing information. US Food and Drug Administration . Accessed December 14, 2023. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/213674s010,203...
-
- Xtandi summary of product characteristics. European Medicines Agency . Accessed December 14, 2023. https://ec.europa.eu/health/documents/community-register/2018/2018020813...
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

