Genomic Insights into Vector-Pathogen Adaptation in Haemaphysalis longicornis and Rhipicephalus microplus
- PMID: 40333071
- PMCID: PMC12030188
- DOI: 10.3390/pathogens14040306
Genomic Insights into Vector-Pathogen Adaptation in Haemaphysalis longicornis and Rhipicephalus microplus
Abstract
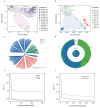
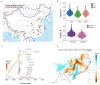
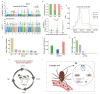
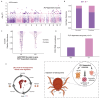
As crucial vectors that transmit pathogens to humans and livestock, ticks pose substantial global health threats and economic burdens. We analyzed 328 tick genomes to explore the population's genetic structure and the adaptive evolution of H. longicornis and R. microplus, two tick species with distinct life cycle characteristics. We observed distinct genetic structures in H. longicornis and R. microplus. Gene flow estimation revealed a closer genetic connection in R. microplus than H. longicornis, which was facilitated by geographical proximity. Notably, we identified a set of candidate genes associated with possible adaptations. Specifically, the immune-related gene DUOX and the iron transport gene ACO1 showed significant signals of natural selection in R. microplus. Similarly, H. longicornis exhibited selection in pyridoxal-phosphate-dependent enzyme genes associated with heme synthesis. Moreover, we observed significant correlations between the abundance of pathogens, such as Rickettsia and Francisella, and specific tick genotypes, which highlights the role of R. microplus in maintaining these pathogens and its adaptations that influence immune responses and iron metabolism, suggesting potential coevolution between vectors and pathogens. Our study highlights the vital genes involved in tick blood feeding and immunity, and it provides insights into the coevolution of ticks and tick-borne pathogens.
Keywords: Haemaphysalis longicornis; Rhipicephalus microplus; tick-borne disease; vector–pathogen adaptation.
Conflict of interest statement
The authors declare that there are no existing competing interests.
Figures
References
-
- Guglielmone A.A., Robbins R.G., Apanaskevich D.A., Petney T.N., Estrada-Peña A., Horak I.G., Shao R., Barker S.C. The Argasidae, Ixodidae and Nuttalliellidae (Acari: Ixodida) of the World: A List of Valid Species Names. Zootaxa. 2010;2528:1. doi: 10.11646/zootaxa.2528.1.1. - DOI
MeSH terms
Grants and funding
- 2023YFC2605400/National Key Research and Development Program of China
- 32288101, 32030020/National Natural Science Foundation of China
- 32470649/National Natural Science Foundation of China
- 23JS141010/Science and Technology Commission of Shanghai Muninicipality
- None/the Office of Global Partnerships (Key Projects Development Fund)
LinkOut - more resources
Full Text Sources
Research Materials

