A Phase 3 Randomized Trial Investigating the Safety, Tolerability, and Immunogenicity of V116, an Adult-Specific Pneumococcal Vaccine, Compared with PPSV23, in Adults ≥50 Years of Age (STRIDE-10)
- PMID: 40333240
- PMCID: PMC12031205
- DOI: 10.3390/vaccines13040341
A Phase 3 Randomized Trial Investigating the Safety, Tolerability, and Immunogenicity of V116, an Adult-Specific Pneumococcal Vaccine, Compared with PPSV23, in Adults ≥50 Years of Age (STRIDE-10)
Abstract
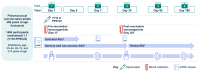
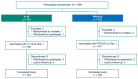
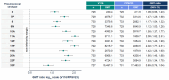
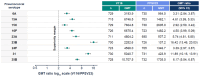
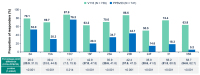
Background: V116 is a 21-valent pneumococcal conjugate vaccine (PCV) designed for adults. It contains the most prevalent serotypes associated with invasive pneumococcal disease (IPD) in adults in regions with established pediatric vaccination programs. This Phase 3 study compared the safety, tolerability, and immunogenicity of V116 with 23-valent pneumococcal polysaccharide vaccine (PPSV23) in adults ≥50 years of age. Methods: In this randomized, active comparator-controlled, parallel-group, multisite, double-blind study, participants were randomized 1:1 to receive a single dose of V116 or PPSV23 (NCT05569954). Primary immunogenicity outcomes assessed the opsonophagocytic activity (OPA) responses for (i) non-inferiority for 12 serotypes common to V116 and PPSV23 based on V116/PPSV23 geometric mean titers (GMTs) at Day 30, and (ii) superiority for nine serotypes unique to V116 using V116/PPSV23 GMTs and the proportions of participants with a ≥4-fold rise in OPA responses from baseline to 30 days post-vaccination. The primary safety outcome was evaluated as the proportion of participants with solicited injection-site and systemic adverse events through Day 5 post-vaccination, and vaccine-related serious adverse events up to 6 months post-vaccination. Findings: V116 was non-inferior to PPSV23 for all 12 common serotypes, superior to PPSV23 for all nine unique serotypes based on V116/PPSV23 GMTs, and superior to PPSV23 for eight of the nine serotypes unique to V116, based on the proportion of participants with a ≥4-fold rise in OPA responses (except for serotype 15C). The safety profile of V116 was comparable to that of PPSV23. Interpretation: In regions with established vaccination programs, V116 could broaden the serotype coverage for residual pneumococcal disease in adults.
Keywords: adult; invasive pneumococcal disease; opsonophagocytic activity; pneumococcal conjugate vaccine; serotype.
Conflict of interest statement
V.Jag. is an employee of MSD (UK) Limited, London, UK. V. Jott. and F.W. are employees of MSD (Switzerland). K.H., C.C., Y.Z., D.F., J.L., U.K.B., and H.P. are employees of Merck Sharp & Dohme LLC (a subsidiary of Merck & Co., Inc., Rahway, NJ, USA). All may own stock and/or hold stock options in Merck & Co., Inc., Rahway, NJ, USA. A.A.D., J.D.V., A.L., S.N.P., R.C., Y.C., O.D., K-H.P., and S.U. have no relevant conflicts of interest to disclose. Merck Sharp & Dohme LLC (a subsidiary of Merck & Co., Inc., Rahway, NJ, USA) funded and participated in the study design, data collection, data analysis, data interpretation, and writing of the report.
Figures

References
-
- Centers for Disease Control and Prevention Active Bacterial Core Surveillance (ABCs) Report: Streptococcus pneumoniae. [(accessed on 16 March 2025)];2019 Available online: https://www.cdc.gov/abcs/downloads/SPN_Surveillance_Report_2019.pdf.
-
- Ramirez J.A., Wiemken T.L., Peyrani P., Arnold F.W., Kelley R., Mattingly W.A., Nakamatsu R., Pena S., Guinn B.E., Furmanek S.P., et al. Adults hospitalized with pneumonia in the United States: Incidence, epidemiology, and mortality. Clin. Infect. Dis. 2017;65:1806–1812. doi: 10.1093/cid/cix647. - DOI - PubMed
-
- GBD Lower Respiratory Infections Collaborators Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect. Dis. 2018;18:1191–1210. doi: 10.1016/S1473-3099(18)30310-4. - DOI - PMC - PubMed
-
- Sabra A., Bourgeois M., Blanc E., Fievez S., Moisi J., Goussiaume G., Lemaitre M., Watier L., Coulombel N., Trehony J., et al. Hospital Burden of All-Cause Pneumonia and Nonbacteremic Pneumococcal Pneumonia in Adults in France Between 2013 and 2019. Open Forum Infect. Dis. 2024;11:ofae349. doi: 10.1093/ofid/ofae349. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Medical

