COX-2 inhibition as a therapeutic strategy for bone loss in Staphylococcus aureus osteomyelitis
- PMID: 40335904
- PMCID: PMC12057237
- DOI: 10.1186/s10020-025-01202-9
COX-2 inhibition as a therapeutic strategy for bone loss in Staphylococcus aureus osteomyelitis
Abstract
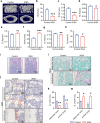
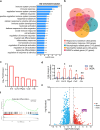
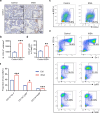
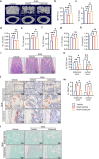
Bone loss in Staphylococcus aureus (S. aureus) osteomyelitis poses a serious challenge to orthopedic treatment, but the underlying mechanism of systemic osteoporosis caused by chronic infection is not completely clear. In this study, γ-irradiation-killed S. aureus (IKSA) was applied to simulate the inflammation and explore the mechanism of systemic bone loss caused by it. In this study, we found that the systemic application of IKSA caused bone loss in mice through increasing osteoclasts and decreasing osteoblasts. An immune response profile with up-regulated COX-2 is identified based on our transcriptional data from IKSA mice bone marrow cells. COX-2 expression is widely up-regulated in bone marrow immune cells, such as myeloid-derived suppressor cells (MDSCs), neutrophils and macrophages in the IKSA-treated mice. Mechanistically, COX-2 stimulated the increasing proportion of MDSCs and neutrophils and the inflammatory response of the bone marrow immune cells, that may regulate bone metabolism. Importantly, COX-2 inhibitor, celecoxib could rescue the bone loss induced by IKSA, which may reason from decrease of inflammatory gene expression in MDSCs, neutrophils and macrophages. Excitingly, COX-2 expression is also increased in bone marrow from mice and patients with S. aureus osteomyelitis. These findings suggested a therapeutic potential for inhibiting COX-2 in combating bone loss in S. aureus osteomyelitis.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The research was ethically sanctioned by the Ethics Committee at Nanfang Hospital. Each participant in this research provided informed consent before taking part. (Approval Number: NFEC-2020-074). All procedures involving animals received approval from the Institutional Animal Care and Use Committee of Nanfang Hospital and were conducted according to the guidelines of the National Regulations on the Management of Laboratory Animals. (Approval Number: NFYY-2022-01037). Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures




References
-
- Arfeen M, Srivastava A, Srivastava N, Khan RA, Almahmoud SA, Mohammed HA. Design, classification, and adverse effects of NSAIDs: a review on recent advancements. Bioorg Med Chem. 2024;112: 117899. 10.1016/j.bmc.2024.117899. - PubMed
MeSH terms
Substances
Grants and funding
- 2022A0007/the President Foundation of Nanfang Hospital, Southern Medical University
- 202412121060/College Students' Innovative Entrepreneurial Training Plan Program
- 82302791/the National Natural Science Foundation Youth Project
- 202201011339/the Science and Technology Project of Guangzhou
- 2022YFC2504305/the National Key Research and Development Program of China
LinkOut - more resources
Full Text Sources
Medical
Research Materials

