Ethylene glycol metabolism in the oleaginous yeast Rhodotorula toruloides
- PMID: 40338313
- PMCID: PMC12062128
- DOI: 10.1007/s00253-025-13504-3
Ethylene glycol metabolism in the oleaginous yeast Rhodotorula toruloides
Abstract
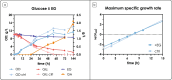
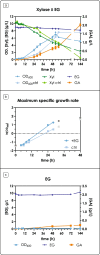
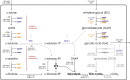
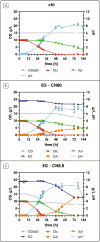
The agro-food chain produces an impressive amount of waste, which includes not only lignocellulosic biomass, but also plastic, used for both protective films and packaging. Thanks to advances in enzymatic hydrolysis, it is now possible to imagine an upcycling that valorizes each waste through microbial fermentation. With this goal in mind, we first explored the ability of the oleaginous red yeast Rhodotorula toruloides to catabolize ethylene glycol (EG), obtained by the hydrolysis of polyethylene terephthalate (PET), in the presence of glucose in batch bioreactor experiments. Secondly, we focused on the physiology of EG catabolism in the presence of xylose as a sole carbon source, and in a mixture of glucose and xylose. Our results show that EG is metabolized to glycolic acid (GA) in all tested conditions. Remarkably, we report for the first time that the consumption of EG improves xylose bioprocess, possibly alleviating a cofactor imbalance by regenerating NAD(P)H. Consumption of EG in the presence of glucose started after the onset of the nitrogen limitation phase, while no significant differences were observed with the control; a 100% mol mol-1 yield of GA was obtained, which has never been reported for yeasts. Finally, a putative EG oxidative pathway was proposed by in silico analyses supported with the existing omics data. Our results propose R. toruloides as a promising candidate for the production of GA from EG that could be exploited simultaneously for the sustainable production of microbial oils from residual hemicellulosic biomasses. KEY POINTS: • Ethylene glycol (EG) is not assimilated as a carbon source by Rhodotorula toruloides • With glucose, EG is oxidized to glycolic acid (GA) with a yield of 100% (mol mol-1) • With xylose, EG to GA is associated with improved growth and xylose uptake rate.
Keywords: Rhodotorula toruloides; Ethylene glycol; Glucose; Glycolic acid; Polyethylene terephthalate; Xylose.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Exploring yeast biodiversity and process conditions for optimizing ethylene glycol conversion into glycolic acid.FEMS Yeast Res. 2024 Jan 9;24:foae024. doi: 10.1093/femsyr/foae024. FEMS Yeast Res. 2024. PMID: 39104224 Free PMC article.
-
Production of D-arabitol from D-xylose by the oleaginous yeast Rhodosporidium toruloides IFO0880.Appl Microbiol Biotechnol. 2018 Jan;102(1):143-151. doi: 10.1007/s00253-017-8581-1. Epub 2017 Nov 11. Appl Microbiol Biotechnol. 2018. PMID: 29127468
-
Valorization of Brewers' Spent Grain for the Production of Lipids by Oleaginous Yeast.Molecules. 2018 Nov 22;23(12):3052. doi: 10.3390/molecules23123052. Molecules. 2018. PMID: 30469531 Free PMC article.
-
Engineering of xylose metabolic pathways in Rhodotorula toruloides for sustainable biomanufacturing.FEMS Yeast Res. 2025 Jan 30;25:foaf029. doi: 10.1093/femsyr/foaf029. FEMS Yeast Res. 2025. PMID: 40498526 Free PMC article. Review.
-
Biotechnological production of glycolic acid and ethylene glycol: current state and perspectives.Appl Microbiol Biotechnol. 2019 Mar;103(6):2525-2535. doi: 10.1007/s00253-019-09640-2. Epub 2019 Feb 1. Appl Microbiol Biotechnol. 2019. PMID: 30707252 Free PMC article. Review.
References
-
- Bonturi N, Crucello A, Viana AJC, Miranda EA (2017) Microbial oil production in sugarcane bagasse hemicellulosic hydrolysate without nutrient supplementation by a Rhodosporidium toruloides adapted strain. Process Biochem 57:16–25. 10.1016/j.procbio.2017.03.007
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources

