RSPO1, a potent inducer of pancreatic β cell neogenesis
- PMID: 40339569
- PMCID: PMC12147903
- DOI: 10.1016/j.xcrm.2025.102126
RSPO1, a potent inducer of pancreatic β cell neogenesis
Abstract
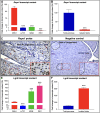
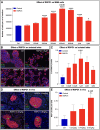
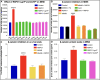
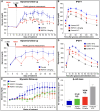
Inducing the neogenesis of pancreatic insulin-producing β cells holds great promise for diabetes research. However, non-toxic compounds with such activities remain to be discovered. Herein, we report the identification of RSPO1, a key agonist of the Wnt/β-catenin pathway, as an inducer of β cell replication. Specifically, we provide evidence that RSPO1 promotes a significant increase in β cell neogenesis in vitro, ex vivo, and in vivo. Importantly, RSPO1 administration is sufficient to activate Wnt/β-catenin signaling in β cells and counter chemically induced or autoimmune-mediated diabetes. Similarly, an optimized analog of RSPO1, allowing for weekly administration, also prevents diabetes in vivo. Lastly, the treatment of transplanted human islets with RSPO1 induces a significant 2.78-fold increase in human β cell numbers in only 60 days, these cells being functional. Such activities of RSPO1 to promote β cell neogenesis could therefore represent an unprecedented hope in the continued search for diabetes alternative therapies.
Keywords: Rspo1; Wnt/β-catenin signaling; diabetes; endocrine pancreas; islets of Langerhans; β cell replication.
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests P.C. is a co-founder and a shareholder of DiogenX. S.S., T.N., C.T., L.E., and L.T. are employees of DiogenX. P.B., J.M., and A.A. are external consultants and shareholders of DiogenX. B.C. is the CEO, co-founder, and shareholder of DiogenX. P.C. and S.S. have a patent related to this work (PCT/EP2021/050289). P.C. and P.B. have a patent related to this work (PCT/EP2022/069925).
Figures



References
-
- El Sayed S.A., Mukherjee S. StatPearls Publishing; 2022. Physiology, Pancreas. - PubMed
-
- Rezania A., Bruin J.E., Arora P., Rubin A., Batushansky I., Asadi A., O’Dwyer S., Quiskamp N., Mojibian M., Albrecht T., et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat. Biotechnol. 2014;32:1121–1133. doi: 10.1038/nbt.3033. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

