Maximizing NMR Sensitivity: A Guide to Receiver Gain Adjustment
- PMID: 40342208
- PMCID: PMC12059252
- DOI: 10.1002/nbm.70046
Maximizing NMR Sensitivity: A Guide to Receiver Gain Adjustment
Abstract
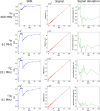
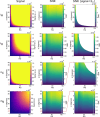
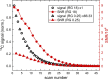
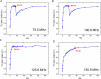
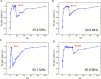
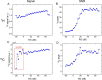
Novel methods and technology drive the rapid advances of nuclear magnetic resonance (NMR). The primary objective of developing novel hardware is to improve sensitivity and reliability (and possibly reduce cost). Automation has made NMR much more convenient, but it may lead to trusting the algorithms without regular checks. In this contribution, we analyzed the signal and signal-to-noise ratio (SNR) as a function of the receiver gain (RG) for 1H, 2H, 13C, and 15N nuclei on five spectrometers. On a 1 T benchtop spectrometer (Spinsolve, Magritek), the SNR showed the expected increase as a function of RG. Still, the 1H and 13C signal amplitudes deviated by up to 50% from supposedly RG-independent signal intensities. On 7, 9.4, 11.7, and 14.1 T spectrometers (Avance Neo, Bruker), the signal intensity increases linearly with RG as expected, but surprisingly, a drastic drop of SNR is observed for some X-nuclei and fields. For example, while RG = 18 provided a 13C SNR similar to that at a maximum RG of 101 at 9.4 T, at RG = 20.2, the determined SNR was 32% lower. The SNR figures are strongly system and resonance frequency dependent. Our findings suggest that NMR users should test the specific spectrometer behavior to obtain optimum SNR for their experiments, as automatic RG adjustment does not account for the observed SNR characteristics. In addition, we provide a method to estimate optimal settings for thermally and hyperpolarized samples of a chosen concentration, polarization, and flip angle, which provide a high SNR and avoid ADC-overflow artefacts.
© 2025 The Author(s). NMR in Biomedicine published by John Wiley & Sons Ltd.
Figures

Similar articles
-
Application of high-field NMR spectroscopy for characterization and quantitation of submilligram quantities of isolated natural products.Magn Reson Chem. 2015 Dec;53(12):1043-50. doi: 10.1002/mrc.4304. Epub 2015 Aug 20. Magn Reson Chem. 2015. PMID: 26289113 Free PMC article.
-
Exploring signal-to-noise ratio and sensitivity in non-uniformly sampled multi-dimensional NMR spectra.J Biomol NMR. 2013 Feb;55(2):167-78. doi: 10.1007/s10858-012-9698-2. Epub 2012 Dec 29. J Biomol NMR. 2013. PMID: 23274692 Free PMC article.
-
Increase in SNR for 31P MR spectroscopy by combining polarization transfer with a direct detection sequence.Magn Reson Med. 2012 Aug;68(2):353-7. doi: 10.1002/mrm.23260. Epub 2011 Dec 12. Magn Reson Med. 2012. PMID: 22162118
-
RotoMate: An open-source, 3D printed autosampler for use with benchtop nuclear magnetic resonance spectrometers.HardwareX. 2021 Jun 23;10:e00211. doi: 10.1016/j.ohx.2021.e00211. eCollection 2021 Oct. HardwareX. 2021. PMID: 35607663 Free PMC article.
-
Benchtop nuclear magnetic resonance spectroscopy in forensic chemistry.Magn Reson Chem. 2023 Feb;61(2):106-129. doi: 10.1002/mrc.5197. Epub 2021 Aug 3. Magn Reson Chem. 2023. PMID: 34286862 Review.
References
-
- Sapir G., Steinberg D. J., Aqeilan R. I., and Katz‐Brull R., “Real‐Time Non‐Invasive and Direct Determination of Lactate Dehydrogenase Activity in Cerebral Organoids—A New Method to Characterize the Metabolism of Brain Organoids?,” Pharmaceuticals 14, no. 9 (2021): 878, 10.3390/ph14090878. - DOI - PMC - PubMed
-
- Sharma R. and Sharma A., “21.1 Tesla Magnetic Resonance Imaging Apparatus and Image Interpretation: First Report of a Scientific Advancement,” Recent Patents on Medical Imaging 1, no. 2 (2011): 89–105.
-
- Kovacs H., Moskau D., and Spraul M., “Cryogenically Cooled Probes—A Leap in NMR Technology,” Progress in Nuclear Magnetic Resonance Spectroscopy 46, no. 2 (2005): 131–155, 10.1016/j.pnmrs.2005.03.001. - DOI
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

