Inhibiting synovial inflammation and promoting cartilage repair in rheumatoid arthritis using a matrix metalloproteinase-binding hydrogel
- PMID: 40343163
- PMCID: PMC12059346
- DOI: 10.1016/j.mtbio.2025.101792
Inhibiting synovial inflammation and promoting cartilage repair in rheumatoid arthritis using a matrix metalloproteinase-binding hydrogel
Abstract
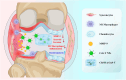
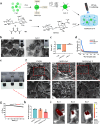
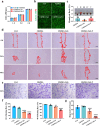
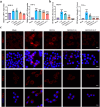
Originating from synovial tissue, matrix metalloproteinase-9 (MMP-9) is a key inflammatory factor that promotes the formation and invasion of synovial pannus, leading to cartilage matrix destruction in rheumatoid arthritis (RA). However, clinical trials of systemic use of MMP-9 inhibitors are not successful due to severe side effects. Thus, locally inhibiting MMP-9 may be an alternative in the treatment of RA. Herein, we developed MMP-9 binding peptide-functionalized copper sulfide nanoparticles (CuS-T NPs) and delivered them with light crosslinking chondroitin sulfate methacrylate (ChSMA) hydrogel. We found that the CuS NP-doped hydrogels could inhibit synovial inflammation. Specifically, the CuS-T/ChSMA hydrogel could rapidly bind to MMP-9, thereby inhibiting not only the invasion of RA fibroblast-like synoviocytes but also the polarization of inflammatory M1-type macrophages. The underlying mechanism involved the inhibition of the MAPK pathway. Moreover, ChSMA hydrogel provided a cartilage matrix-mimic microenvironment and synergistically promoted the generation of collagen-2 and aggrecan with CuS NPs. In an adjuvant-induced arthritis mouse model, the intra-articular injection of ChSMA/CuS-T hydrogel significantly alleviated synovial inflammation and accelerated cartilage repair without causing any side effects, killing two birds with one stone in RA therapy.
Keywords: Chondroitin sulfate methacrylate; Copper sulfide nanoparticles; Matrix metalloproteinase-9; Rheumatoid arthritis.
© 2025 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
Endogenous MMP-9 and not MMP-2 promotes rheumatoid synovial fibroblast survival, inflammation and cartilage degradation.Rheumatology (Oxford). 2014 Dec;53(12):2270-9. doi: 10.1093/rheumatology/keu254. Epub 2014 Jun 29. Rheumatology (Oxford). 2014. PMID: 24982240
-
Turnover of type II collagen and aggrecan in cartilage matrix at the onset of inflammatory arthritis in humans: relationship to mediators of systemic and local inflammation.Arthritis Rheum. 2003 Nov;48(11):3085-95. doi: 10.1002/art.11331. Arthritis Rheum. 2003. PMID: 14613270
-
Self-actuating inflammation responsive hydrogel microsphere formulation for controlled drug release in rheumatoid arthritis (RA): Animal trials and study in human fibroblast like synoviocytes (hFLS) of RA patients.Biomater Adv. 2024 Jun;160:213853. doi: 10.1016/j.bioadv.2024.213853. Epub 2024 Apr 16. Biomater Adv. 2024. PMID: 38636119
-
Advanced application of stimuli-responsive drug delivery system for inflammatory arthritis treatment.Mater Today Bio. 2022 Feb 21;14:100223. doi: 10.1016/j.mtbio.2022.100223. eCollection 2022 Mar. Mater Today Bio. 2022. PMID: 35243298 Free PMC article. Review.
-
Wnt signaling pathway in rheumatoid arthritis, with special emphasis on the different roles in synovial inflammation and bone remodeling.Cell Signal. 2013 Oct;25(10):2069-78. doi: 10.1016/j.cellsig.2013.04.002. Epub 2013 Apr 17. Cell Signal. 2013. PMID: 23602936 Review.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

