PDGF-BB overexpressing dental pulp stem cells improve angiogenesis in dental pulp regeneration
- PMID: 40343206
- PMCID: PMC12058851
- DOI: 10.3389/fbioe.2025.1578410
PDGF-BB overexpressing dental pulp stem cells improve angiogenesis in dental pulp regeneration
Abstract
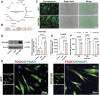
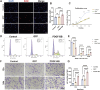
Introduction: Angiogenesis represents a critical challenge in dental pulp regeneration due to the tissue's restricted nutrient supply through a 0.5-mm apical foramen. While dental pulp stem cells (DPSCs) hold regenerative potential, their limited vascularization capacity impedes clinical applications. Through Single-cell RNA sequencing (scRNA-seq) analysis of human dental pulp, we discovered a PDGF (+) mesenchymal subset exhibiting enhanced angiogenic signatures, suggesting targeted cell selection could overcome this bottleneck.
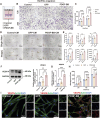
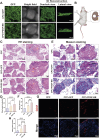
Methods: ScRNA-seq identified PDGF (+) subpopulation in human pulp samples, validated through multiplex immunohistochemical of the localization of PDGF/CD73/CD31. PDGF-BB-overexpressing DPSCs were engineered via lentiviral vectors. Functional assessments included: 1) CCK-8/Edu/cell cycle/transwell assays for proliferation and migration ability 2) HUVECs co-culture models analyzing chemotaxis and tube formation 3) Vascularized tissue formation in rat kidney capsule transplants.
Results and discussion: The CD73 (+) PDGF (+) subpopulation demonstrated spatial correlation with CD31 (+) vasculature. PDGF-BB overexpression enhanced DPSCs' proliferative capacity and migration capacity. Co-cultured HUVECs exhibited increased tube formation with PDGF-BB group. In vivo transplants generated more vascular structures containing CD31 (+) endothelia. These findings establish PDGF-BB engineering as an effective strategy to amplify DPSCs' angiogenic potential, while emphasizing the therapeutic value of functionally-defined stem cell subpopulations in pulp regeneration.
Keywords: dental pulp regeneration; dental pulp stem cells; endothelial; single-cell RNA sequencing; vascularization.
Copyright © 2025 Jiang, Duan, Li, Yan, Si, Xu, Li, Zhang and Gu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The Effects of Platelet-Derived Growth Factor-BB on Human Dental Pulp Stem Cells Mediated Dentin-Pulp Complex Regeneration.Stem Cells Transl Med. 2017 Dec;6(12):2126-2134. doi: 10.1002/sctm.17-0033. Epub 2017 Oct 24. Stem Cells Transl Med. 2017. PMID: 29064632 Free PMC article.
-
Dental pulp stem cells overexpressing stromal-derived factor-1α and vascular endothelial growth factor in dental pulp regeneration.Clin Oral Investig. 2019 May;23(5):2497-2509. doi: 10.1007/s00784-018-2699-0. Epub 2018 Oct 12. Clin Oral Investig. 2019. PMID: 30315421
-
Inhibition of soluble epoxide hydrolase enhances the dentin-pulp complex regeneration mediated by crosstalk between vascular endothelial cells and dental pulp stem cells.J Transl Med. 2024 Jan 16;22(1):61. doi: 10.1186/s12967-024-04863-y. J Transl Med. 2024. PMID: 38229161 Free PMC article.
-
Dental Pulp Stem Cells: Advances to Applications.Stem Cells Cloning. 2020 Feb 13;13:33-42. doi: 10.2147/SCCAA.S166759. eCollection 2020. Stem Cells Cloning. 2020. PMID: 32104005 Free PMC article. Review.
-
Systematic Review of Human Dental Pulp Stem Cells for Cartilage Regeneration.Tissue Eng Part B Rev. 2020 Feb;26(1):1-12. doi: 10.1089/ten.TEB.2019.0140. Epub 2020 Jan 22. Tissue Eng Part B Rev. 2020. PMID: 31744404
References
-
- Cabaña-Muñoz, Eugenia M., Pelaz Fernández M. J., Parmigiani-Cabaña J. M., Parmigiani-Izquierdo J. M., Merino J. J. (2023). Adult mesenchymal stem cells from oral cavity and surrounding areas: types and Biomedical applications. Pharmaceutics 15 (8), 2109. 10.3390/pharmaceutics15082109 - DOI - PMC - PubMed
-
- Caseiro A. R., Santos Pedrosa S., Ivanova G., Vieira Branquinho M., Almeida A., Faria F., et al. (2019). Mesenchymal stem/Stromal cells Metabolomic and bioactive factors profiles: a comparative analysis on the umbilical Cord and dental pulp derived stem/Stromal cells secretome. PloS One 14 (11), e0221378. 10.1371/journal.pone.0221378 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

