Carboprost versus Oxytocin as the first-line treatment of primary postpartum haemorrhage (COPE): protocol for a phase IV, double-blind, double-dummy, randomised controlled trial and economic analysis
- PMID: 40345687
- PMCID: PMC12067827
- DOI: 10.1136/bmjopen-2025-101255
Carboprost versus Oxytocin as the first-line treatment of primary postpartum haemorrhage (COPE): protocol for a phase IV, double-blind, double-dummy, randomised controlled trial and economic analysis
Abstract
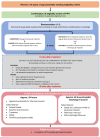
Introduction: Excessive bleeding after childbirth (postpartum haemorrhage, PPH) affects 5% of births and causes 75 000 maternal deaths worldwide annually. It is the leading cause of direct maternal deaths globally and continues to be a major cause of mortality in the UK. Oxytocin is the standard first-line treatment for atonic PPH. The PPH rate is increasing, and this may be partially related to the overuse of oxytocics in labour. Laboratory studies on myometrium suggest that repeated use of oxytocics leads to the saturation of oxytocin receptors and reduced therapeutic efficacy of oxytocin. Carboprost (a prostaglandin analogue) is usually reserved for second-line management of atonic PPH. A systematic review comparing the efficacy of carboprost and conventional uterotonics for PPH prophylaxis found that carboprost was associated with less blood loss, but around 15% of women experienced side effects. The study's aim is to compare intramuscular carboprost with intravenous oxytocin for the initial treatment of PPH. In addition, to assess the cost-effectiveness of both treatments, participants' views on the two treatments and the consent process.
Methods and analysis: COPE is a double-blind, double-dummy, randomised controlled trial that aims to recruit 2000 women (1:1 allocation, stratified by mode of birth) across 20 hospitals in the UK. Due to the emergency nature of PPH, COPE uses a research without prior consent (RWPC) model. Randomisation and treatment will occur if eligibility criteria are met once bleeding starts. Postnatal consent will be sought for disclosure of identifiable data and continued follow-up. Clinical efficacy outcomes will be collected at 24 and 48 hours or at hospital discharge, if sooner. Questionnaires will also be collected at 24 hours and 4 weeks postrandomisation. Cost-effectiveness will be based on the incremental cost per quality-adjusted life-year, calculated from the perspective of the NHS and personal social services.
Ethics and dissemination: This study has been approved by the Coventry and Warwickshire Research Ethics Committee (REC) (18/WM/0227) and the Health Research Authority. Results will be disseminated via peer-reviewed publications.
Trial registration number: ISRCTN16416766.
Keywords: HEALTH ECONOMICS; Maternal medicine; Postpartum Women; Pregnancy; QUALITATIVE RESEARCH; Randomized Controlled Trial.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY. Published by BMJ Group.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- World Health Organization . Geneva: World Health Organization; 2023. A roadmap to combat postpartum haemorrhage between 2023 and 2030. Licence: CC BY-NC-SA 3.0 IGO.
-
- Knight MN, Tuffnell D, Shakespeare J, et al. Saving lives, improving mothers’ care - Lessons learned to inform maternity care from the UK and Ireland confidential enquiries into maternal deaths and morbidity 2013–15. Oxford: National Perinatal Epidemiology Unit, University of Oxford; 2017.
-
- Intrapartum care. NICE guideline. NG235. 2023
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous