Effects of empagliflozin on functional capacity, LV filling pressure, and cardiac reserves in patients with type 2 diabetes mellitus and heart failure with preserved ejection fraction: a randomized controlled open-label trial
- PMID: 40346546
- PMCID: PMC12065317
- DOI: 10.1186/s12933-025-02756-y
Effects of empagliflozin on functional capacity, LV filling pressure, and cardiac reserves in patients with type 2 diabetes mellitus and heart failure with preserved ejection fraction: a randomized controlled open-label trial
Abstract
Background: Clinical trials have established the prognostic benefits of sodium‒glucose cotransporter 2 (SGLT2) inhibitors in patients with type 2 diabetes mellitus (T2DM) and heart failure (HF) with preserved ejection fraction (HFpEF), although the underlying mechanisms are not clearly understood. The purpose of this study was to determine the effects of the SGLT2 inhibitor empagliflozin on functional capacity, left ventricular (LV) diastolic function/filling pressure, and cardiac reserves in patients with HFpEF and T2DM.
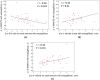
Methods: In the present prospective single-center trial, we enrolled 70 diabetic patients with stable HF according to the New York Heart Association functional class II-III criteria, an LV ejection fraction ≥ 50%, and increased LV filling pressure at rest and/or during exercise (determined by echocardiography). The patients were randomly assigned in an open-label fashion to the empagliflozin group (10 mg a day, n = 35) or the control group (n = 35) for 6 months. Echocardiography (at rest and during exercise), the 6-min walk test distance (6MWD), blood levels of N-terminal pro-brain natriuretic peptide (NT-proBNP), and the profibrotic biomarker sST2 were analysed at baseline and 6 months after randomization. The primary endpoint was the change in the 6MWD, and the secondary endpoints included the change in the left atrial (LA) volume index, early mitral inflow to mitral annulus relaxation velocity (E/e') ratio both at rest and during exercise, key cardiac reserves and biomarkers in the blood from baseline to 6 months.
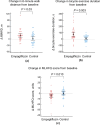
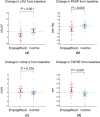
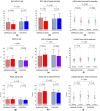
Results: After 6 months of empagliflozin therapy, the 6MWTD significantly increased, whereas the LA volume index and the E/e' ratio both at rest and during exercise decreased compared with those of the control group (P < 0.05 for all). LV diastolic, LA reservoir and contractile, and chronotropic reserves also improved in the empagliflozin group compared with those in the control group (P < 0.05 for all). Furthermore, treatment with empagliflozin led to improvements in NT-proBNP and ST2 blood levels compared with those in the control group (P < 0.05 for both).
Conclusions: In diabetic patients with HFpEF, empagliflozin treatment improved exercise capacity, which appeared to be the result of favourable effects on LV diastolic dysfunction and key cardiac reserves: LV diastolic, LA reservoir and contractile, and chronotropic. These haemodynamic mechanisms may underline the benefits of SGLT2 inhibitors in large-scale HFpEF trials.
Trial registration: URL: https://www.
Clinicaltrials: gov . Unique Identifier NCT03753087.
Keywords: Cardiac reserve; Diastolic dysfunction; Empagliflozin; Heart failure with preserved ejection fraction; Type 2 diabetes.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study complied with the Declaration of Helsinki and was approved by the Ethics Committee of the Institute of Clinical Cardiology (Moscow, Russia). All patients provided written informed consent before study enrollment. All the data were anonymized to prevent any potential breaches of patient privacy. Consent for publication: All the authors provided consent for the publication of the article. Competing interests: The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- 2018-9-25/A grant from the Ministry of Health of the Russian Federation
- 2018-9-25/A grant from the Ministry of Health of the Russian Federation
- 2018-9-25/A grant from the Ministry of Health of the Russian Federation
- 2018-9-25/A grant from the Ministry of Health of the Russian Federation
- 2018-9-25/A grant from the Ministry of Health of the Russian Federation
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

