Calcium channels in anesthesia management: A molecular and clinical review
- PMID: 40346957
- PMCID: PMC12163254
- DOI: 10.1177/17448069251343417
Calcium channels in anesthesia management: A molecular and clinical review
Abstract
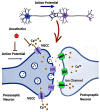
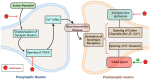
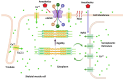
Calcium channels play an essential role in the molecular and physiological mechanisms underlying anesthesia by mediating intracellular calcium ion (Ca2+) flux, which regulates key processes such as neurotransmitter release, neuronal excitability, and immune responses. Voltage-gated calcium channels (VGCCs) and ligand-gated calcium channels (LGCCs) are integral to the anesthetic process, with subtypes such as T-type VGCCs and NMDA receptors influencing consciousness and pain perception. This review emphasizes current evidence to highlight how anesthetic agents interact with calcium channels via direct inhibition and modulation of intracellular signaling pathways, such as phosphatidylinositol metabolism. Additionally, calcium channelopathies - genetic or acquired dysfunctions affecting VGCCs and LGCCs - pose challenges in anesthetic management, including arrhythmias, malignant hyperthermia, and altered anesthetic sensitivity. These findings underscore the critical need for precision medicine approaches tailored to patients with these conditions. While significant progress has been made in understanding the roles of calcium channels in anesthesia, knowledge gaps remain regarding the long-term implications of anesthetic interactions on calcium signaling and clinical outcomes. This review bridges foundational science with clinical practice, emphasizing the translational potential of calcium channel research for optimizing anesthetic strategies. By integrating molecular insights with emerging pharmacogenomic approaches, it provides a pathway for developing safer and more effective anesthesia protocols that enhance patient outcomes.
Keywords: Anesthesia; anesthetic mechanisms; calcium channelopathies; calcium channels.
Conflict of interest statement
Declaration of conflicting interestsThe author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
Similar articles
-
The role of voltage-gated calcium channels in the mechanisms of anesthesia and perioperative analgesia.Curr Opin Anaesthesiol. 2022 Aug 1;35(4):436-441. doi: 10.1097/ACO.0000000000001159. Epub 2022 Jul 5. Curr Opin Anaesthesiol. 2022. PMID: 35787588 Free PMC article. Review.
-
Are neuronal voltage-gated calcium channels valid cellular targets for general anesthetics?Channels (Austin). 2010 Nov-Dec;4(6):518-22. doi: 10.4161/chan.4.6.12873. Epub 2010 Nov 1. Channels (Austin). 2010. PMID: 21164281 Free PMC article. Review.
-
Voltage-gated calcium channels: Determinants of channel function and modulation by inorganic cations.Prog Neurobiol. 2015 Jun;129:1-36. doi: 10.1016/j.pneurobio.2014.12.003. Epub 2015 Mar 25. Prog Neurobiol. 2015. PMID: 25817891 Review.
-
Short-Term Facilitation at a Detonator Synapse Requires the Distinct Contribution of Multiple Types of Voltage-Gated Calcium Channels.J Neurosci. 2017 May 10;37(19):4913-4927. doi: 10.1523/JNEUROSCI.0159-17.2017. Epub 2017 Apr 14. J Neurosci. 2017. PMID: 28411270 Free PMC article.
-
Therapeutic potential of Voltage gated Calcium channels.Mini Rev Med Chem. 2008 Oct;8(12):1285-90. doi: 10.2174/138955708786141016. Mini Rev Med Chem. 2008. PMID: 18855741 Review.
References
-
- Fullick W, Fullick J. Organs, systems, and surgery. Oxford: Oxford University Press, 2023.
-
- Pankratov Y, Lalo U. Calcium permeability of ligand-gated Ca2+ channels. Eur J Pharmacol 2014; 739: 60–73. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

