EXCALIBER-RRMM: a phase III trial of iberdomide, daratumumab, and dexamethasone in relapsed/refractory multiple myeloma
- PMID: 40346992
- PMCID: PMC12150652
- DOI: 10.1080/14796694.2025.2501920
EXCALIBER-RRMM: a phase III trial of iberdomide, daratumumab, and dexamethasone in relapsed/refractory multiple myeloma
Abstract
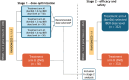
Multiple myeloma (MM) is a plasma cell neoplasm that stems from the malignant transformation of clonal plasma cells. It is characterized by multiple periods of remission and relapse requiring multiple lines of therapy, with response to treatment and survival decreasing with each successive relapse. To achieve deep and durable responses in relapsed/refractory MM (RRMM), novel treatments are required. Iberdomide (IBER), an oral CELMoD™ agent, is associated with greater tumoricidal and immune-stimulatory effects than immunomodulatory drugs (IMiDs®). IBER has been shown to have synergy with dexamethasone (DEX) and daratumumab (DARA) in vitro. In a phase I/II trial, IBER in combination with DARA and DEX (IberDd) was well tolerated and demonstrated promising preliminary efficacy in patients with heavily pretreated RRMM, including patients refractory to IMiD agents, DARA, and proteasome inhibitors. EXCALIBER-RRMM is a unique confirmatory phase III trial that incorporates a 2-stage seamless design to firstly address dose optimization of IberDd, and secondly, to compare the efficacy and safety of the selected IberDd dose with DARA, bortezomib, and DEX in patients with early-line (1 or 2 prior lines of therapy) RRMM.Clinical trial registration: www.clinicaltrials.gov identifier is NCT04975997.
Keywords: CELMoD agent; Relapsed/refractory multiple myeloma; clinical trial; iberdomide; phase III.
Plain language summary
Multiple myeloma (MM) is a type of cancer that affects a group of blood cells called plasma cells, found in the bone marrow, which become abnormal and grow uncontrollably. Eventually, these cells crowd out healthy blood cells and cause various problems in the body, including bone pain, tiredness, weakness, and frequent infections. A cancer is referred to as “relapsed” when the disease reappears or grows again after a period of remission, and as “refractory” when it does not respond to, or stops responding to, treatment. While treatment options for MM have continued to get better, unfortunately MM is still not curable, and there is a need for new treatment options to prevent it from relapsing and to provide long-term control of the disease. Researchers wanted to see whether adding a new treatment called iberdomide (IBER) to a combination of established treatments can stop relapsed/refractory MM from getting worse and help patients live longer. To answer this question, they have started a unique large, international, phase III study called EXCALIBER-RRMM comprising two stages. The first stage will select the appropriate dose of IBER (dose optimization), and the second stage will evaluate whether treating patients with the selected dose of IBER in combination with daratumumab (DARA) and dexamethasone (DEX), compared with a combination of DARA, bortezomib, and DEX, can help stop the cancer from getting worse and help patients live longer.
Conflict of interest statement
Professional medical writing and editorial assistance were provided by Mauro Locati, PhD, of Excerpta Medica, and was funded by Bristol Myers Squibb. The authors are fully responsible for all content and editorial decisions.
Figures

References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
