Intermediate Filament Protein BFSP1 Maintains Oocyte Asymmetric Division by Modulating Spindle Length
- PMID: 40349178
- PMCID: PMC12302563
- DOI: 10.1002/advs.202504066
Intermediate Filament Protein BFSP1 Maintains Oocyte Asymmetric Division by Modulating Spindle Length
Abstract
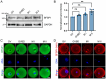
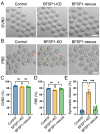
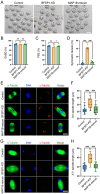
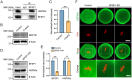
The cytoskeleton is composed of microtubules, microfilaments, and intermediate filaments in cells. While the functions of microtubules and microfilaments have been well elucidated, the roles of intermediate filaments and associated proteins remain largely unknown, especially in meiosis. BFSP1 is an intermediate filament protein mainly expressed in the eye lens to play important roles in the development of congenital cataract. Here, we document that BFSP1 functions as a spindle regulator to drive the oocyte asymmetric division. Specifically, we found that BFSP1 distributed on the spindle apparatus during oocyte meiotic maturation. Depletion of BFSP1 resulted in symmetric division of oocytes, accompanied by the formation of elongated spindles at metaphase I and anaphase/telophase I stages. In addition, immunoprecipitation combined with mass spectrometry analysis identified MAP1B, a microtubule-associated protein, as an interacting partner of BFSP1. Depletion or mutation of MAP1B phenocopied the meiotic defects observed in BFSP1-depleted oocytes, and expression of exogenous MAP1B-EGFP in BFSP1-depleted oocytes recovered the spindle length and asymmetric division. We further determined that BFSP1 recruited molecular chaperone HSP90α on the spindle to stabilize MAP1B, thereby controlling the spindle length. To sum up, our findings reveal a unique meiotic role for BFSP1 in the regulation of spindle dynamics and oocyte asymmetric division.
Keywords: BFSP1; asymmetric division; intermediate filament protein; oocyte meiosis; spindle length.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
CLASP1 regulates DYNC1I1 for PLK1-mediated spindle organization and cytokinesis in oocyte meiosis.J Cell Sci. 2025 Jul 15;138(14):jcs264015. doi: 10.1242/jcs.264015. Epub 2025 Jul 22. J Cell Sci. 2025. PMID: 40501366
-
Intermediate Filament Protein BFSP2 Controls Spindle Formation via HSC70-Mediated Stabilization of CLTC During Oocyte meiosis.Adv Sci (Weinh). 2025 Jul 2:e06639. doi: 10.1002/advs.202506639. Online ahead of print. Adv Sci (Weinh). 2025. PMID: 40600490
-
The doublecortin-family kinase ZYG-8DCLK1 regulates microtubule dynamics and motor-driven forces to promote the stability of C. elegans acentrosomal spindles.PLoS Genet. 2024 Sep 3;20(9):e1011373. doi: 10.1371/journal.pgen.1011373. eCollection 2024 Sep. PLoS Genet. 2024. PMID: 39226307 Free PMC article.
-
Molecular Guardians of Oocyte Maturation: A Systematic Review on TUBB8, KIF11, and CKAP5 in IVF Outcomes.Int J Mol Sci. 2025 Jul 2;26(13):6390. doi: 10.3390/ijms26136390. Int J Mol Sci. 2025. PMID: 40650169 Free PMC article. Review.
-
Oocyte activation for women following intracytoplasmic sperm injection (ICSI).Cochrane Database Syst Rev. 2024 Dec 20;12(12):CD014040. doi: 10.1002/14651858.CD014040.pub2. Cochrane Database Syst Rev. 2024. PMID: 39704318
References
-
- Parekh S. H., Chaudhuri O., Theriot J. A., Fletcher D. A., Nat. Cell Biol. 2005, 7, 1219. - PubMed
-
- Gao J., Nakamura F., J. Biomed Res. 2024, 39, 20240193.
-
- Romet‐Lemonne G., Leduc C., Jegou A., Wioland H., Annu. Rev. Biophys. 2025. - PubMed
-
- Coelho‐Rato L. S., Parvanian S., Andrs Salajkova S., Medalia O., Eriksson J. E., J. Cell Sci. 2024, 137, jcs261386. - PubMed
-
- Kechagia Z., Eibauer M., Medalia O., Curr. Opin. Cell Biol. 2024, 89, 102375. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
