Fat traffic control: S-acylation in axonal transport
- PMID: 40349611
- PMCID: PMC12264562
- DOI: 10.1016/j.molpha.2025.100039
Fat traffic control: S-acylation in axonal transport
Abstract
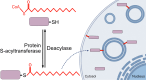
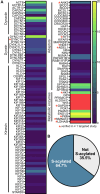
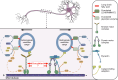
Neuronal axons serve as a conduit for the coordinated transport of essential molecular cargo between structurally and functionally distinct subcellular compartments via axonal molecular machinery. Long-distance, efficient axonal transport of membrane-bound organelles enables signal transduction and neuronal homeostasis. Efficient axonal transport is conducted by dynein and kinesin ATPase motors that use a local ATP supply from metabolic enzymes tethered to transport vesicles. Molecular motor adaptor proteins promote the processive motility and cargo selectivity of fast axonal transport. Axonal transport impairments are directly causative or associated with many neurodegenerative diseases and neuropathologies. Cargo specificity, cargo-adaptor proteins, and posttranslational modifications of cargo, adaptor proteins, microtubules, or the motor protein subunits all contribute to the precise regulation of vesicular transit. One posttranslational lipid modification that is particularly important in neurons in regulating protein trafficking, protein-protein interactions, and protein association with lipid membranes is S-acylation. Interestingly, many fast axonal transport cargos, cytoskeletal-associated proteins, motor protein subunits, and adaptors are S-acylated to modulate axonal transport. Here, we review the established regulatory role of S-acylation in fast axonal transport and provide evidence for a broader role of S-acylation in regulating the motor-cargo complex machinery, adaptor proteins, and metabolic enzymes from low-throughput studies and S-acyl-proteomic data sets. We propose that S-acylation regulates fast axonal transport and vesicular motility through localization of the proteins required for the motile cargo-complex machinery and relate how perturbed S-acylation contributes to transport impairments in neurological disorders. SIGNIFICANCE STATEMENT: This review investigates the regulatory role of S-acylation in fast axonal transport and its connection to neurological diseases, with a focus on the emerging connections between S-acylation and the molecular motors, adaptor proteins, and metabolic enzymes that make up the trafficking machinery.
Keywords: S-acylation; fast axonal transport; glycolysis; molecular motor proteins; neurological disorders; palmitoylation.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare no conflicts of interest.
Figures
Similar articles
-
Using Live-Cell Imaging to Measure the Effects of Pathological Proteins on Axonal Transport in Primary Hippocampal Neurons.J Vis Exp. 2023 Dec 22;(202):10.3791/66156. doi: 10.3791/66156. J Vis Exp. 2023. PMID: 38189521 Free PMC article.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
The gE/gI complex is necessary for kinesin-1 recruitment during alphaherpesvirus egress from neurons.J Virol. 2025 Jan 31;99(1):e0165024. doi: 10.1128/jvi.01650-24. Epub 2024 Dec 9. J Virol. 2025. PMID: 39651860 Free PMC article.
-
S-acylation in apoptotic and non-apoptotic cell death: a central regulator of membrane dynamics and protein function.Biochem Soc Trans. 2025 Apr 29;53(2):487-96. doi: 10.1042/BST20253012. Biochem Soc Trans. 2025. PMID: 40304073 Free PMC article. Review.
-
Effects of consumers and health providers working in partnership on health services planning, delivery and evaluation.Cochrane Database Syst Rev. 2021 Sep 15;9(9):CD013373. doi: 10.1002/14651858.CD013373.pub2. Cochrane Database Syst Rev. 2021. PMID: 34523117 Free PMC article.
Cited by
-
Novel Lactams as SARM1 Inhibitors for Treating Axonal Degeneration.ACS Med Chem Lett. 2025 May 9;16(6):931-932. doi: 10.1021/acsmedchemlett.5c00241. eCollection 2025 Jun 12. ACS Med Chem Lett. 2025. PMID: 40529072
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

