From function to structure: how myofibrillogenesis influences the transverse-axial tubular system development and its peculiarities
- PMID: 40352140
- PMCID: PMC12062141
- DOI: 10.3389/fphys.2025.1576133
From function to structure: how myofibrillogenesis influences the transverse-axial tubular system development and its peculiarities
Abstract
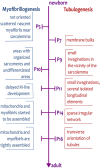
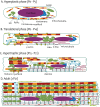
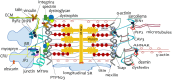
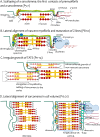
The transverse-axial tubular system (TATS) is the extension of sarcolemma growing to the cell interior, providing sufficient calcium signaling to induce calcium release from sarcoplasmic reticulum cisternae and stimulate the contraction of neighboring myofibrils. Interestingly, the development of TATS is delayed and matures during the post-partum period. It starts with small invaginations near the sarcolemma, proceeding to grow an irregular network that is later assembled into the notably transversally oriented tubular network. Accumulating evidence supports the idea that the development of TATS is linked to cell dimensions, calcium signaling, and increasing myofibrillar content orchestrated by electromechanical stimulation. However, the overall mechanism has not yet been described. The topic of this review is the development of TATS with an emphasis on the irregular phase of tubule growth. The traditional models of BIN1-related tubulation are also discussed. We summarized the recently described protein interactions during TATS development, mainly mediated by costameric and sarcomeric proteins, supporting the idea of the coupling sites between TATS and the myofibrils. We hypothesize that the formation and final organization of the tubular system is driven by the simultaneous development of the contractile apparatus under cycling electromechanical stimulus.
Keywords: Z-line; cardiomyocyte; costameres; myofibrillogenesis; postnatal development; sarcomere; t-tubules; transverse–axial tubular system.
Copyright © 2025 Sevcikova Tomaskova and Mackova.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Ampong B. N., Imamura M., Matsumiya T., Yoshida M., Takeda S. (2005). Intracellular localization of dysferlin and its association with the dihydropyridine receptor. Acta Myol. 24, 134–144. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

