Comparative Efficacy of Ciltacabtagene Autoleucel Versus Standard-of-Care Treatments for Patients with Previously Treated Relapsed or Refractory Multiple Myeloma: A Matching-Adjusted Indirect Comparison
- PMID: 40354013
- PMCID: PMC12182486
- DOI: 10.1007/s12325-025-03205-8
Comparative Efficacy of Ciltacabtagene Autoleucel Versus Standard-of-Care Treatments for Patients with Previously Treated Relapsed or Refractory Multiple Myeloma: A Matching-Adjusted Indirect Comparison
Abstract
Introduction: Matching adjusted indirect comparisons (MAICs) were performed to compare the efficacy of cilta-cel versus elotuzumab + pomalidomide + dexamethasone (EloPd), isatuximab + carfilzomib + dexamethasone (IsaKd), isatuximab + pomalidomide + dexamethasone (IsaPd), and selinexor + bortezomib + dexamethasone (SVd) in patients with relapsed or refractory multiple myeloma (RRMM) who have received at least one prior therapy and are lenalidomide-refractory.
Methods: Unanchored MAICs were performed using individual patient-level data (IPD) for all apheresed patients randomized to the cilta-cel arm of CARTITUDE-4 (n = 208) and published arm-level data for EloPd from ELOQUENT-3 (n = 60), IsaKd from IKEMA (lenalidomide-refractory subgroup, n = 57), IsaPd from ICARIA-MM (n = 154), and SVd from BOSTON (lenalidomide-refractory subgroup, n = 53). Eligibility criteria from each comparator trial were applied to the cilta-cel arm IPD, and further imbalances in patient characteristics were adjusted by weighting the cilta-cel patient data to match the reported baseline characteristics of the comparator trials. Comparative efficacy was estimated for overall response rate, very good partial response or better (≥ VGPR) rate, complete response or better (≥ CR) rate, progression-free survival (PFS), and overall survival (OS).
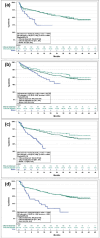
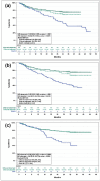
Results: After adjustment, cilta-cel patients were significantly more likely to achieve an overall response versus EloPd, IsaPd, and SVd, and were significantly more likely to achieve ≥ VGPR and ≥ CR versus all comparators. Cilta-cel patients also had significant reductions in the risk of disease progression or death (PFS) versus all comparators: 64% versus EloPd, 49% versus IsaKd, 69% versus IsaPd, and 62% versus SVd. Similarly, cilta-cel patients had significant improvements in OS for all feasible comparisons: 52% versus EloPd, 58% versus IsaPd, and 60% versus SVd.
Conclusion: Cilta-cel patients demonstrated clinically meaningful benefits over EloPd, IsaKd, IsaPd, and SVd for response and survival outcomes, highlighting its superiority over alternative treatment options for patients with RRMM who have received at least one prior therapy and are refractory to lenalidomide.
Keywords: Ciltacabtagene autoleucel; Matching-adjusted indirect comparison; Multiple myeloma.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of Interest: Noemi Puig: Amgen: Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; Sanofi: Membership on an entity's Board of Directors or advisory committees; Pfizer: Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; The Binding Site: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau. Joris Diels, Suzy van Sanden, João Mendes, Seina Lee, Teresa Hernando, Jordan M. Schecter, and Nikoletta Lendvai: Employees of Janssen and shareholders of Johnson & Johnson. Nitin Patel: Employee and shareholder of Legend Biotech USA Inc., has divested equity with BMS (via spouse). Heather Burnett and Allie Cichewicz: Employees of Evidera, a Thermo Fisher Company which provides consulting and other research services to life science companies. In their salaried positions, they work with a variety of companies and is precluded from receiving payment or honoraria directly from these organizations for services rendered. Evidera received payment from Johnson & Johnson for the conduct of this study. José María Sanchez-Pina: Consultancy and Speakers Bureau Johnson and Johnson, Gilead, Amgen, SanofiSerena Rocchi: Nothing to disclose. Roberto Mina: Received honoraria from AbbVie, Amgen, Bristol Myers Squibb, Celgene, GSK, Janssen, Sanofi, Takeda, Menarini Stemline and Pfizer; participated in advisory boards for Amgen, Bristol Myers Squibb, GSK, Janssen, Sanofi and Pfizer. Paolo Corradini: Honoraria and travel received from: Abbvie, Amgen, BeiGene, BMS, Eli Lilly, Gilead/Kite, GSK, Incyte, Janssen, Jazz Pharma, Novartis, Pfizer, Roche, Sanofi, SOBI, Takeda. Michele Cavo: Served as a consultant and received travel grants from Janssen. Jesus San Miguel: declares participation on advisory boards and consulting services, on behalf of my Institution, for Abbvie, Amgen, BMS, Celgene, GSK, Haemalogix, Janssen-Cilag, Karyopharm, MSD, Novartis, Pfizer, Takeda, Regeneron, Roche, Sanofi, SecuraBio and Gilead-Kite. Leyla Shune: serves as on J&J advisory boards. Abdullah M. Khan: Received research funding from BMS and Sanofi; member of the Speakers Bureau for Amgen, BMS, and Sanofi. Surbhi Sidana: Has received Research Funding: Magenta Therapeutics, BMS, Allogene, Janssen, Novartis and serves as a consultant for: Magenta Therapeutics, BMS, Janssen, Sanofi, Takeda, Regeneron, Abbvie, Pfizer, BiolineRx, Legend, Genentech, Arcellx, Kite. Xavier Leleu: Kite, A Gilead Company: Consultancy, Honoraria. Salomon Manier: Research funding Janssen, Celgene/BMS; Consultant for Abbvie, Adaptive Biotechnology, Amgen, Celgene/BMS, GlaxoSmithKline, Janssen, Novartis, Pfizer, Regeneron, Roche, Sanofi, Takeda; Travel funding: Abbvie, Adaptive Biotechnology, Amgen, Celgene/BMS, GlaxoSmithKline, Janssen, Novartis, Pfizer, Regeneron, Roche, Sanofi. Brea Lipe: Consulting: Janssen, Sanofi, Karyopharm, BMS, Pfizer, GSK and Research: Amgen, Karyopharm. Katja Weisel: Research Grant: Abbvie, Amgen, BMS/Celgene, GSK, Janssen, Sanofi (to the Institution). Honoraria: Abbvie, Amgen, Adaptive Biotech, Astra Zeneca, Beigene, BMS, Celgene, Janssen, GSK, Karyopharm, Menarini, Novartis, Oncopeptides, Pfizer, Roche, Sanofi, Stemline, Takeda; Adisory Board: Abbvie, Amgen, Adaptive Biotech, Beigene, BMS, Celgene, Janssen, GSK, Karyopharm, Menarini, Novartis, Oncopeptides, Pfizer, Regeneron, Roche, Sanofi, Takeda. Joaquin Martinez-Lopez: has stock ownership in altum, statb therapeutics; has received honorarium from Johnson and Johnson, BMS, Pfizer, Incity, Roche, Sanofi, Novartis; has served as a consultant for Johnson and Johnson, BMS, Pfizer, Incity, Roche, Sanofi, Novartis; has served on Speaker’s Bureau for Johnson and Johnson, BMS, Pfizer, Incity, Roche, Sanofi, Novartis, Amgen; has research funding from BMS, J&J, Incity, Pfizer, Amgen and travel funds from Johnson and Johnson, BMS, Pfizer, Incity, Roche, Sanofi, Novartis. Ethical Approval: This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. This analysis utilized de-identified data from published clinical trial data and therefore ethics committee approval was not required. Each individual trial included in this analysis was approved by independent ethics committees or institutional review boards at each study site and all patients provided written informed consent before enrolling in each clinical trial. Study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments.
Figures
Similar articles
-
Patient-reported outcomes following ciltacabtagene autoleucel or standard of care in patients with lenalidomide-refractory multiple myeloma (CARTITUDE-4): results from a randomised, open-label, phase 3 trial.Lancet Haematol. 2025 Jan;12(1):e45-e56. doi: 10.1016/S2352-3026(24)00320-X. Lancet Haematol. 2025. PMID: 39756844 Clinical Trial.
-
Comparative Effectiveness of Ciltacabtagene Autoleucel in CARTITUDE-4 Versus Real-World Physician's Choice of Therapy from the Flatiron Registry in Lenalidomide-Refractory Multiple Myeloma.Adv Ther. 2025 Aug 6. doi: 10.1007/s12325-025-03308-2. Online ahead of print. Adv Ther. 2025. PMID: 40768190
-
Survival Outcomes with Cilta-cel Versus Conventional Treatment Regimens for Patients with Lenalidomide-Refractory Multiple Myeloma Using Inverse Probability of Treatment Weighting.Adv Ther. 2025 Sep;42(9):4418-4431. doi: 10.1007/s12325-025-03278-5. Epub 2025 Jul 4. Adv Ther. 2025. PMID: 40613875 Clinical Trial.
-
Systematic Literature Review and Network Meta-Analysis of Treatment Outcomes in Relapsed and/or Refractory Multiple Myeloma.J Clin Oncol. 2017 Apr 20;35(12):1312-1319. doi: 10.1200/JCO.2016.71.1663. Epub 2017 Feb 27. J Clin Oncol. 2017. PMID: 28240968
-
The clinical effectiveness and cost-effectiveness of bortezomib and thalidomide in combination regimens with an alkylating agent and a corticosteroid for the first-line treatment of multiple myeloma: a systematic review and economic evaluation.Health Technol Assess. 2011 Dec;15(41):1-204. doi: 10.3310/hta15410. Health Technol Assess. 2011. PMID: 22146234 Free PMC article.
References
-
- Kumar S, Paiva B, Anderson KC, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17(8):e328–46. - PubMed
-
- Avet-Loiseau H, et al. Poster EP1008 Treatment patterns of lenalidomide delivery and outcomes in multiple myeloma patients in real-world settings: a multi-center retrospective study. 25th Annual EHA Congress; June 11–21, 2020; Virtual2020.
-
- Hajek et al. 3204 Patient characteristics and survival outcomes of lenalidomide exposed non-refractory vs. lenalidomide refractory multiple myeloma patients in the HONEUR federated data network. 64th American Society of Hematology Annual Meeting and Exposition 2022 (ASH 2022); 10–13 December 2022; New Orleans, Louisiana2022.

