Efficacy and safety outcomes of the Paul glaucoma implant compared to the Ahmed glaucoma valve
- PMID: 40360615
- PMCID: PMC12075782
- DOI: 10.1038/s41598-025-00839-0
Efficacy and safety outcomes of the Paul glaucoma implant compared to the Ahmed glaucoma valve
Abstract
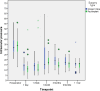
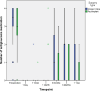
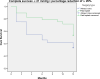
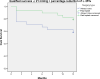
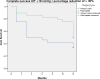
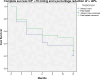
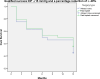
This study compares the one-year outcomes of standalone Ahmed glaucoma valve (AGV) implantation and standalone Paul glaucoma implant (PGI) in adult patients with primary and secondary glaucoma. A retrospective, single-center, comparative study was conducted on adult patients who underwent standalone PGI and AGV at the University Medical Center Mainz. The primary outcome measures were the changes of IOP and the number of antiglaucoma eye medication at one year postoperatively. Secondary outcome measures included complete and qualified success rates, failure rates, visual acuity logMAR and the incidence of adverse events. A total of 24 adult patients were included in the AGV group and 28 in the PGI group. The median preoperative intraocular pressure decreased from 29.5mmHg (Interquartile range (IQR) 21-42) to 16.0mmHg (IQR 7-37) in the AGV group, and from 34.0 mmHg (IQR 13-56) to 16.0 (IQR 7-21) mmHg in the PGI group at the one-year follow-up. The median number of classes of intraocular pressure-lowering medications reduced from 3.5 to 0 in the AGV group, and from 3.0 to 0 in the PGI group. There were no statistically significant differences between the groups for any success criteria or failure. The AGV produced more encapsulation than the PGI, and the latter more tube exposures. Both the Ahmed Glaucoma Valve and the Paul Glaucoma Implant effectively reduce IOP and the number of antiglaucoma medications at one year with comparable safety profiles.
Keywords: Ahmed glaucoma valve; Drainage; Glaucoma; Paul glaucoma implant; Tube.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: EMH has received research support and consultation fees from Thea, Heidelberg Engineering, Santen, Ora, Allergan, Meyetech, and Belkin Laser. Additionally, Professor Hoffmann has served on advisory boards for these companies.AKS received research support by Bayer, Heidelberg Engineering, Novartis and Santen, and acted as consultant for Abbvie and Santen.JR served as a consultant for Heidelberg Engineering and SantenAll the remaining authors declare no conflict of interest.
Figures

References
-
- Khodeiry, M. & Sayed, M. S. New glaucoma drainage implants available to glaucoma surgeons. Curr. Opin. Ophthalmol.34, 176–180 (2023). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

