Fecal Arachidonic Acid: A Potential Biomarker for Inflammatory Bowel Disease Severity
- PMID: 40362272
- PMCID: PMC12071911
- DOI: 10.3390/ijms26094034
Fecal Arachidonic Acid: A Potential Biomarker for Inflammatory Bowel Disease Severity
Abstract
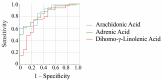
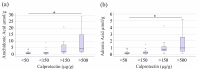
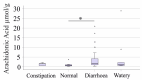
Arachidonic acid levels are elevated in the colonic mucosa of patients with inflammatory bowel disease (IBD). Fecal metabolites are emerging as valuable diagnostic tools for IBD. This study aimed to investigate associations between 31 fecal fatty acids, including arachidonic acid, to identify potential correlations with disease severity. Among the 31 fatty acids analyzed in feces, dihomo-γ-linolenic acid, arachidonic acid, and adrenic acid were significantly increased in patients with IBD compared to controls. In contrast, levels of linoleic acid and γ-linolenic acid, the precursors of arachidonic acid, were similar between both groups. No significant differences in fatty acid levels were observed between patients with Crohn's disease and ulcerative colitis. Arachidonic acid and adrenic acid levels positively correlated with fecal calprotectin, a clinically established marker of IBD severity, but showed no association with stool consistency or the Gastrointestinal Symptom Rating Scale. This suggests that these fatty acids are linked to disease severity rather than disease-related symptoms. Current IBD-specific medications had no significant impact on the fecal levels of any of the 31 fatty acids. In summary, this study demonstrates elevated fecal levels of dihomo-γ-linolenic acid, arachidonic acid, and adrenic acid in IBD patients. Normal levels of precursor fatty acids suggest that impaired downstream metabolism may contribute to the accumulation of these n-6 polyunsaturated fatty acids.
Keywords: biomarker; calprotectin; fatty acids; feces; inflammatory bowel disease; stool consistency.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

