Role of FHOD1 in tumor cells and tumor immune microenvironment
- PMID: 40364836
- PMCID: PMC12069282
- DOI: 10.3389/fimmu.2025.1514488
Role of FHOD1 in tumor cells and tumor immune microenvironment
Abstract
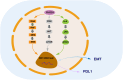
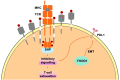
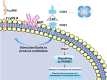
FHOD 1 (Formin homology 2 domain containing protein 1) is a member of Diaphanous-related formins (DRFs) which contains a GTP-binding domain (GBD), formin homology (FH) 1 and FH 2 domains, a coiled-coil, and a diaphanous-like autoregulatory domain. Studies have shown that FHOD1 can not only regulate intracellular signals in tumor cells but also regulate various components of the tumor microenvironment (TME), such as T cells, B cells, cancer-associated fibroblasts (CAFs), some cytokines. Aberrant expression and dysfunction of the FHOD1 protein play a key role in tumor immunosuppression. Specifically, FHOD1 can impair function of chemokine receptors that are supposed to direct immune cells to localize to the tumor site accurately. As a result of this impairment, immune cells cannot migrate efficiently into TME, thereby impairing their ability to attack tumor cells. In addition, FHOD1 activated signaling pathways within the immune cells abnormally, resulting in their inability to recognize and destroy tumor cells effectively. Therefore, FHOD1 ultimately leads to a state of immunosuppression in TME, providing favorable conditions for the growth and spread of tumor cells. Altogether this review provides an in-depth understanding of the role of FHOD1 in tumor immunosuppression.
Keywords: FHOD1; PD-1; PDL1; epithelial-mesenchymal transition; tumor immune microenvironment.
Copyright © 2025 Yang, Kang, Cai, Jia, Fan and Zhu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Biochemical characterization of the diaphanous autoregulatory interaction in the formin homology protein FHOD1.J Biol Chem. 2006 Feb 24;281(8):5084-93. doi: 10.1074/jbc.M509226200. Epub 2005 Dec 16. J Biol Chem. 2006. PMID: 16361249
-
The human formin FHOD1 contains a bipartite structure of FH3 and GTPase-binding domains required for activation.Structure. 2008 Sep 10;16(9):1313-23. doi: 10.1016/j.str.2008.06.008. Structure. 2008. PMID: 18786395
-
Structures of FHOD1-Nesprin1/2 complexes reveal alternate binding modes for the FH3 domain of formins.Structure. 2021 Jun 3;29(6):540-552.e5. doi: 10.1016/j.str.2020.12.013. Epub 2021 Jan 19. Structure. 2021. PMID: 33472039 Free PMC article.
-
FHOD proteins in actin dynamics--a formin' class of its own.Small GTPases. 2014;5(2):11. doi: 10.4161/21541248.2014.973765. Small GTPases. 2014. PMID: 25483300 Free PMC article. Review.
-
Formins as effector proteins of Rho GTPases.Small GTPases. 2014;5:e29513. doi: 10.4161/sgtp.29513. Epub 2014 Jun 10. Small GTPases. 2014. PMID: 24914801 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

