Clinical and Biological Remission With Tezepelumab: The Real-World Response in Severe Uncontrolled Asthma
- PMID: 40365686
- PMCID: PMC12186586
- DOI: 10.1111/all.16590
Clinical and Biological Remission With Tezepelumab: The Real-World Response in Severe Uncontrolled Asthma
Abstract
Introduction: Tezepelumab is an anti-TSLP monoclonal antibody approved for the treatment of severe asthma. It has broad downstream anti-T2 effects, offering the prospect of biological remission. Real-world data on clinical remission rates with tezepelumab is lacking, and the relationship between clinical and biological remission is unclear. Finally, the effectiveness of tezepelumab in patients who have failed to respond to existing biologic therapies is unknown.
Methods: Clinical and biomarker data from adults with severe asthma treated with tezepelumab in a real-world setting was analyzed. Clinical outcome measures including clinical remission were recorded along with rates of biological remission (defined as blood eosinophil count < 300 cells/mcL and FeNO < 25 ppb).
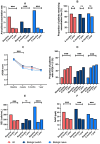
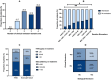
Results: One hundred seventy-five patients were included. 98/175 (56%) had switched from another biologic. Following tezepelumab initiation, the exacerbation rate decreased from 3.1 (2.5) to 0.8 (1.4), with 59% of patients remaining exacerbation-free at 1 year. 54% achieved an ACQ score < 1.5. Clinical remission at 1 year was observed in 36%, with a rate of 55% in T2-high patients versus 19% in T2 low patients. The clinical response in biologic-naïve and biologic switch patients was similar. FeNO declined from 41 ppb (24-76) to 24 ppb (16-38) and BEC fell from 300 cells/μL (60-610) to 180 cells/μL (105-320) (both p < 0.001). 38% achieved biological remission. 15% attained both clinical and biological remission.
Conclusion: Tezepelumab led to substantial clinical improvements and clinical remission in up to 55% of T2-high patients with severe asthma. A disconnect between clinical and biological remission was observed. The long-term significance of residual T2 inflammation on tezepelumab is unknown.
Keywords: asthma; asthma treatment; biologics.
© 2025 The Author(s). Allergy published by European Academy of Allergy and Clinical Immunology and John Wiley & Sons Ltd.
Conflict of interest statement
J.G. reports payment or honoraria for lectures and educational events from Astra Zeneca and Sanofi. A.M.N. reports payment or honoraria for lectures and educational events from Astra Zeneca, Glaxo Smith Kline, and Sanofi and support for attending conferences from Astra Zeneca, Chiesi, and Napp. L.T. reports payment or honoraria for lectures and educational events and travel support from Astra Zeneca and Teva. GdA reports advisory board work for, been engaged to provide training to HCPs on behalf of, received research grants from, and/or been sponsored to attend an educational meeting by: AstraZeneca, Boehringer‐Ingleheim, Chiesi, GlaxoSmithKline, NAPP, Novartis, Pfizer, Sanofi, Teva. J.D. reports support to attend conferences from Sanofi. C.R. reports honoraria for lectures and conference support from Astra Zeneca. D.J.J. reports research grants to the department from Astra Zeneca, consulting fees from Astra Zeneca, GSK, and Sanofi Regeneron, payments or honoraria for lectures and educational events from Astra Zeneca, GSK, and Sanofi.FC is a recipient of the European Academy of Allergy and Clinical Immunology (EAACI) Research Fellowship 2024 and has received honoraria from Astra Zeneca, GSK, Sanofi, Chiesi, and Doxa Pharma. L.G., M.F., J.L.L., T.M., F.H., and P.K. report no COI.
Figures
References
-
- Bleecker E. R., FitzGerald J. M., Chanez P., et al., “Efficacy and Safety of Benralizumab for Patients With Severe Asthma Uncontrolled With High‐Dosage Inhaled Corticosteroids and Long‐Acting β2‐Agonists (SIROCCO): A Randomised, Multicentre, Placebo‐Controlled Phase 3 Trial,” Lancet 388, no. 10056 (2016): 2115–2127. - PubMed
-
- Busse W. W., Maspero J. F., Rabe K. F., et al., “Liberty Asthma QUEST: Phase 3 Randomized, Double‐Blind, Placebo‐Controlled, Parallel‐Group Study to Evaluate Dupilumab Efficacy/Safety in Patients With Uncontrolled, Moderate‐To‐Severe Asthma,” Advances in Therapy 35, no. 5 (2018): 737–748, 10.1007/s12325-018-0702-4. - DOI - PMC - PubMed
-
- FitzGerald J. M., Bleecker E. R., Nair P., et al., “Benralizumab, an Anti‐Interleukin‐5 Receptor α Monoclonal Antibody, as Add‐On Treatment for Patients With Severe, Uncontrolled, Eosinophilic Asthma (CALIMA): A Randomised, Double‐Blind, Placebo‐Controlled Phase 3 Trial,” Lancet 388, no. 10056 (2016): 2128–2141. - PubMed
-
- Pavord I. D., Korn S., Howarth P., et al., “Mepolizumab for Severe Eosinophilic Asthma (DREAM): A Multicentre, Double‐Blind, Placebo‐Controlled Trial,” Lancet 380, no. 9842 (2012): 651–659. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

