The comparative effectiveness and safety of fluticasone-salmeterol via metered-dose versus dry powder inhalers for COPD: A new user cohort study
- PMID: 40367410
- PMCID: PMC12077913
- DOI: 10.1371/journal.pmed.1004596
The comparative effectiveness and safety of fluticasone-salmeterol via metered-dose versus dry powder inhalers for COPD: A new user cohort study
Abstract
Background: Fluticasone-salmeterol is available in both metered-dose and dry powder inhaler formulations for the treatment of chronic obstructive pulmonary disease (COPD). Metered-dose inhalers are associated with substantially higher greenhouse gas emissions than dry powder inhalers; however, data on their comparative effectiveness and safety in COPD remain limited. We aimed to compare the effectiveness and safety of fluticasone-salmeterol delivered via metered-dose inhaler (Advair HFA) versus dry powder inhaler (Advair Diskus) among patients with COPD treated in routine care.
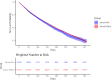
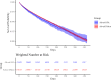
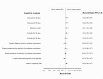
Methods and findings: We conducted a retrospective cohort study using Optum's de-identified Clinformatics DataMart (January 1, 2007 to November 30, 2023). The study included 202,052 commercially insured patients aged 40 years or older with COPD who had continuous insurance coverage for 180 days prior to cohort entry and had not initiated any inhaled corticosteroid-long-acting β₂-agonist during that period. Patients receiving fluticasone-salmeterol via a metered-dose inhaler (exposure) were compared to those receiving these drugs via a dry powder inhaler (referent), with stabilized inverse probability of treatment weighting applied for covariate adjustment. The primary effectiveness outcome was the incidence of first moderate or severe COPD exacerbation within 365 days of cohort entry. The primary safety outcome was the incidence of first pneumonia hospitalization during the same period. Use of fluticasone-salmeterol via metered-dose inhaler was associated with a similar hazard of first moderate or severe COPD exacerbation (hazard ratio [HR], 1.03; 95% confidence interval [CI], 0.99 to 1.08) and first pneumonia hospitalization (HR, 1.06; 95% CI, 0.98 to 1.14) compared to the dry powder inhaler. Primary study limitations include potential residual confounding despite weighting and short follow-up times.
Conclusions: In this cohort study comparing two brand-name fluticasone-salmeterol inhalers prescribed for COPD in routine clinical practice, effectiveness and safety outcomes were similar for patients receiving metered-dose and dry powder versions.
Copyright: © 2025 Demkowicz et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: BJD reports employment for Health Advances, a private healthcare strategy consulting firm. SVW reports ad hoc consulting for Exponent Inc and MITRE, a federally funded research and development center for the Centers for Medicare and Medicaid. ASK serves on the editorial board of PLOS Medicine. ASK and WBF report serving as consultants for Alosa Health. WBF has served as an expert witness in litigation against inhaler manufacturers.
Figures


Similar articles
-
Budesonide-Formoterol Metered-Dose Inhaler vs Fluticasone-Salmeterol Dry-Powder Inhaler.JAMA Intern Med. 2025 Aug 1;185(8):1005-1013. doi: 10.1001/jamainternmed.2025.2299. JAMA Intern Med. 2025. PMID: 40622686 Free PMC article.
-
Comparative Effectiveness and Safety of Generic Versus Brand-Name Fluticasone-Salmeterol to Treat Chronic Obstructive Pulmonary Disease.Ann Intern Med. 2023 Aug;176(8):1047-1056. doi: 10.7326/M23-0615. Epub 2023 Aug 8. Ann Intern Med. 2023. PMID: 37549393 Free PMC article. Clinical Trial.
-
The comparative effectiveness of initiating fluticasone/salmeterol combination therapy via pMDI versus DPI in reducing exacerbations and treatment escalation in COPD: a UK database study.Int J Chron Obstruct Pulmon Dis. 2017 Aug 17;12:2445-2454. doi: 10.2147/COPD.S141409. eCollection 2017. Int J Chron Obstruct Pulmon Dis. 2017. PMID: 28860739 Free PMC article.
-
Fluticasone Propionate/Salmeterol MDPI (AirDuo RespiClick®): A Review in Asthma.Clin Drug Investig. 2018 May;38(5):463-473. doi: 10.1007/s40261-018-0644-2. Clin Drug Investig. 2018. PMID: 29582249 Review.
-
An innovative corticosteroid/long-acting β2-agonist breath-triggered inhaler: facilitating lung delivery of fluticasone propionate/formoterol fumarate for the treatment of asthma.Expert Opin Drug Deliv. 2019 Dec;16(12):1367-1380. doi: 10.1080/17425247.2019.1689957. Epub 2019 Nov 28. Expert Opin Drug Deliv. 2019. PMID: 31752560 Review.
References
-
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: 2024 report [Internet]. Global Initiative for Chronic Obstructive Lung Disease; 2024. [cited 2024 Jul 24]. Available from: https://goldcopd.org/2024-gold-report/.
-
- Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention: 2024 report [Internet]. Global Initiative for Asthma; 2024. [cited 2024 Jul 24]. Available from: https://ginasthma.org/2024-report/.
-
- Rabin AS, Harlan EA, Ambinder AJ. Small devices, big problems: addressing the global warming potential of metered-dose inhalers. Ann Am Thorac Soc [Internet]. 2022. Jul [cited 2024 Mar 24];19(7):1090–2. Available from: https://www.atsjournals.org/doi/10.1513/AnnalsATS.202202-131VP. - DOI - PMC - PubMed
-
- National Institute for Health and Care Excellence (NICE). Patient decision aid: inhalers for asthma. London, United Kingdom: National Institute for Health and Care Excellence; 2020.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

