Canagliflozin alleviates progestin resistance by suppressing RARβ/CRABP2 signaling in THRB knockout endometrial cancer cells
- PMID: 40371351
- PMCID: PMC12075957
- DOI: 10.3389/fphar.2025.1573032
Canagliflozin alleviates progestin resistance by suppressing RARβ/CRABP2 signaling in THRB knockout endometrial cancer cells
Abstract
Introduction: Progestin resistance has emerged as a significant barrier to the conservative management of endometrial cancer (EC). The mechanisms underlying progestin resistance in endocrine therapy remain incompletely understood. Previous studies have suggested that silencing thyroid hormone receptor B (THRB) is associated with progestin resistance in EC cells.
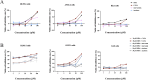
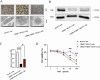
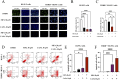
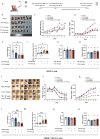
Methods: THRB-knockout RL95-2 (THRB(-/-)/RL95-2) cells were constructed to investigate progestin resistance mechanisms. Cell proliferation and apoptosis were assessed in RL95-2 and THRB(-/-)/RL95-2 cells treated with canagliflozin (CANA), medroxyprogesterone acetate (MPA), and their combination using CCK-8, EdU, and flow cytometry assays. In vivo, nude mouse xenograft models were used to evaluate the efficacy of CANA and MPA. Transcriptomic and proteomic analyses were performed to identify pathways associated with progestin resistance. Molecular dynamics simulations, along with western blotting and immunohistochemistry were utilized to validate the targets of CANA. Electrophoretic mobility shift assays and dual luciferase reporter assays were employed to investigate the interactions between TRβ, RARβ, and CRABP2.
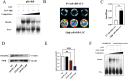
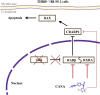
Results: THRB(-/-)/RL95-2 cells were successfully constructed. CANA demonstrated a strong binding affinity for TRβ. Both MPA and CANA suppressed proliferation in RL95-2 cells, but MPA was ineffective in THRB(-/-)/RL95-2 cells, indicating that THRB deficiency induced progestin resistance. CANA significantly inhibited proliferation and promoted apoptosis in THRB(-/-)/RL95-2 cells. In vivo, CANA, either alone or in combination with MPA, significantly reduced tumor growth in xenograft models derived from both wild-type and THRB-knockout RL95-2 cells. Transcriptomic and proteomic analyses revealed that progestin resistance in EC was linked to the retinoic acid signaling pathways. Western blotting confirmed that the expressions of RARβ, RXRA and CRABP2 were significantly elevated in THRB(-/-)/RL95-2 cells. Treatment with CANA, alone or in combination with MPA, effectively reduced the expression of these proteins. Immunohistochemical analysis demonstrated that RARβ expression was significantly increased in uterine tissues from patients with progestin-insensitive EC or endometrial atypical hyperplasia. Electrophoretic mobility shift assays and dual luciferase reporter assays demonstrated that TRβ negatively modulated RARβ expression by binding to its promoter, while RARβ positively regulated CRABP2 expression.
Conclusion: THRB knockout activated retinoic acid pathway, leading to progestin resistance. CANA targeted RARβ and RXRA, downregulated CRABP2, restored BAX levels, and counteracted progestin resistance. The combination of CANA and MPA presented a novel strategy for alleviating progestin resistance and enhancing clinical efficacy.
Keywords: canagliflozin; endometrial cancer; medroxyprogesterone acetate; progestin resistance; retinoic acid receptor β; thyroid hormone receptor β.
Copyright © 2025 Yang, Zhou, Lv, Ni, Hu, Wang, Qu, Li, Yang, Zhong, Chen and Zhu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
Thyroid Hormone Receptor β Knockdown Reduces Suppression of Progestins by Activating the mTOR Pathway in Endometrial Cancer Cells.Int J Mol Sci. 2022 Oct 19;23(20):12517. doi: 10.3390/ijms232012517. Int J Mol Sci. 2022. PMID: 36293372 Free PMC article.
-
Nomegestrol Acetate Suppresses Human Endometrial Cancer RL95-2 Cells Proliferation In Vitro and In Vivo Possibly Related to Upregulating Expression of SUFU and Wnt7a.Int J Mol Sci. 2017 Jun 22;18(7):1337. doi: 10.3390/ijms18071337. Int J Mol Sci. 2017. PMID: 28640224 Free PMC article.
-
Epidermal growth factor receptor signaling pathway involved in progestin-resistance of human endometrial carcinoma: In a mouse model.J Obstet Gynaecol Res. 2012 Dec;38(12):1358-66. doi: 10.1111/j.1447-0756.2012.01881.x. Epub 2012 May 21. J Obstet Gynaecol Res. 2012. PMID: 22612393
-
Epidermal growth factor receptor signaling enhanced by long-term medroxyprogesterone acetate treatment in endometrial carcinoma.Gynecol Oncol. 2007 Apr;105(1):45-54. doi: 10.1016/j.ygyno.2006.12.014. Epub 2007 Jan 22. Gynecol Oncol. 2007. PMID: 17240435
-
NrCAM secreted by endometrial stromal cells enhances the progestin sensitivity of endometrial cancer cells through epigenetic modulation of PRB.Cancer Gene Ther. 2022 Oct;29(10):1452-1462. doi: 10.1038/s41417-022-00467-0. Epub 2022 Apr 6. Cancer Gene Ther. 2022. PMID: 35388173 Free PMC article.
References
-
- Abdelmaksoud N. M., El-Mahdy H. A., Ismail A., Elsakka E. G. E., El-Husseiny A. A., Khidr E. G., et al. (2023). The role of miRNAs in the pathogenesis and therapeutic resistance of endometrial cancer: a spotlight on the convergence of signaling pathways. Pathol. Res. Pract. 244, 154411. 10.1016/j.prp.2023.154411 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

