Differences in the product characteristics and clinical use of granulocytes for transfusion: The BEST Collaborative study
- PMID: 40371893
- PMCID: PMC12168426
- DOI: 10.1111/trf.18263
Differences in the product characteristics and clinical use of granulocytes for transfusion: The BEST Collaborative study
Abstract
Background: Whether granulocytes for transfusion are beneficial remains uncertain, although some evidence suggests that efficacy may be dose-related. Granulocytes are mostly produced by apheresis procedure, but other means of production are increasingly used.
Methods: Centers that produce and/or use granulocytes were recruited through the BEST Collaborative and completed a detailed survey of granulocyte manufacture, specifications, clinical use, operational considerations, and data collection initiatives.
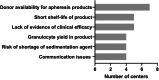
Results: Fifteen national, regional, and local producers and/or users of granulocytes were included. Granulocytes were produced from apheresis procedure (n = 10), pooled buffy coats (n = 2), single buffy coats (n = 4) or pooling of residual leukocyte units from whole blood processing (n = 1). The mean adult dose of granulocytes reported was 1.6 to 3.7 × 1010 for apheresis, and 1.8 to 2.2 × 1010 for pooled buffy coat granulocytes. For apheresis procedure donations, donor stimulation included steroids and/or granulocyte colony-stimulating factor. Centers providing whole blood-derived granulocytes reported shorter times from request to delivery than those using apheresis procedure products. Indications and product selection criteria were similar. The most frequently reported challenges with granulocytes were donor availability for apheresis procedure (n = 7), short shelf life (n = 5) and lack of evidence of efficacy (n = 5). The cost of one unit of apheresis procedure granulocytes ranged from 568 to 7500 PPP-USD, and for one pooled buffy coat unit was from 2208 to 2822 PPP-USD.
Conclusions: We have highlighted differences in granulocyte production that are relevant for the design and interpretation of much needed international clinical studies.
Keywords: blood; buffy coats; granulocytes; transfusion.
© 2025 The Author(s). Transfusion published by Wiley Periodicals LLC on behalf of AABB.
Conflict of interest statement
The authors have disclosed no conflicts of interest. PT and VdlT are employed by Etablissement Français du Sang (EFS), the French transfusion public service in charge of blood donation and blood component manufacturing, testing and issuing in France.
Figures
References
-
- Bashir S, Stanworth S, Massey E, Goddard F, Cardigan R. Neutrophil function is preserved in a pooled granulocyte component prepared from whole blood donations. Br J Haematol. 2008;140:701–711. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources