Leishmania regulates host YY1: Comparative proteomic analysis identifies infection modulated YY1 dependent proteins
- PMID: 40373059
- PMCID: PMC12080872
- DOI: 10.1371/journal.pone.0323227
Leishmania regulates host YY1: Comparative proteomic analysis identifies infection modulated YY1 dependent proteins
Abstract
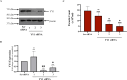
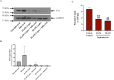
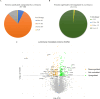
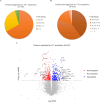
The protein Yin-Yang 1 (YY1) is a ubiquitous multifunctional transcription factor. Interestingly, there are several cellular functions controlled by YY1 that could play a role in Leishmania pathogenesis. Leishmaniasis is a human disease caused by protozoan parasites of the genus Leishmania. This study examined the potential role of macrophage YY1 in promoting Leishmania intracellular survival. Deliberate knockdown of YY1 resulted in attenuated survival of Leishmania in infected macrophages, suggesting a role of YY1 in Leishmania persistence. Biochemical fractionation studies revealed Leishmania infection caused redistribution of YY1 to the cytoplasm from the nucleus where it is primarily located. Inhibition of nuclear transport by leptomycin B attenuates infection-mediated YY1 redistribution and reduces Leishmania survival. This suggests that Leishmania induces the translocation of YY1 from the nucleus to the cytoplasm of infected cells, where it may regulate host molecules to favour parasite survival. A label-free quantitative whole proteome approach showed that the expression of a large number of macrophage proteins was dependent on the YY1 level. Interestingly, several of these proteins were modulated in Leishmania-infected cells, revealing YY1-dependent host response and suggesting their potential role in Leishmania pathogenesis. Together, this study identifies YY1 as a novel virulence factor that promotes Leishmania survival inside host macrophages.
Copyright: © 2025 Brar et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Yin Yang 1: Role in Leishmaniasis.Cells. 2025 Jul 25;14(15):1149. doi: 10.3390/cells14151149. Cells. 2025. PMID: 40801584 Free PMC article. Review.
-
Leishmania infection upregulates and engages host macrophage Argonaute 1, and system-wide proteomics reveals Argonaute 1-dependent host response.Front Immunol. 2023 Nov 30;14:1287539. doi: 10.3389/fimmu.2023.1287539. eCollection 2023. Front Immunol. 2023. PMID: 38098491 Free PMC article.
-
Characterization of Argonaute-containing protein complexes in Leishmania-infected human macrophages.PLoS One. 2024 May 23;19(5):e0303686. doi: 10.1371/journal.pone.0303686. eCollection 2024. PLoS One. 2024. PMID: 38781128 Free PMC article.
-
Leishmania molecules that mediate intracellular pathogenesis.Microbes Infect. 2014 Sep;16(9):721-6. doi: 10.1016/j.micinf.2014.07.012. Epub 2014 Aug 5. Microbes Infect. 2014. PMID: 25107580 Review.
-
Sensing Host Arginine Is Essential for Leishmania Parasites' Intracellular Development.mBio. 2020 Oct 13;11(5):e02023-20. doi: 10.1128/mBio.02023-20. mBio. 2020. PMID: 33051367 Free PMC article.
Cited by
-
Yin Yang 1: Role in Leishmaniasis.Cells. 2025 Jul 25;14(15):1149. doi: 10.3390/cells14151149. Cells. 2025. PMID: 40801584 Free PMC article. Review.
References
-
- Maxfield L, Crane JS. Leishmaniasis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022. [cited 2022 Jul 27]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK531456/
-
- Leishmaniasis [Internet]. [cited 2023 Oct 30]. Available from: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

