Single-Cell RNA-Seq Reveals Aging-Related Impairment of Microglial Efferocytosis Contributing to Apoptotic Cells Accumulation After Retinal Injury
- PMID: 40374315
- PMCID: PMC12341814
- DOI: 10.1111/acel.70097
Single-Cell RNA-Seq Reveals Aging-Related Impairment of Microglial Efferocytosis Contributing to Apoptotic Cells Accumulation After Retinal Injury
Abstract
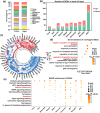
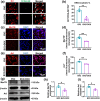
Aging is associated with increased retinal cell apoptosis, which contributes to decreases in retinal function. Apoptotic retinal cell clearance relies on microglial efferocytosis, but the impact of aging on this process has not been fully elucidated. In this study, we aimed to shed light on this by using single-cell RNA sequencing (sc-RNA-seq) to compare young and aged mouse retinal transcriptional profiles, in which 74,412 retinal cells from young and aged mice were classified into 10 transcriptionally distinct retinal cell types, and differentially expressed genes between young versus aged retinas were mainly associated with cellular senescence and apoptosis. Furthermore, ligand-receptor interactions (e.g., AXL-GAS6, MERTK-GAS6) between microglia and other retinal cells were strengthened in aged, compared to young retinas. Additionally, among microglia, Subcluster 4 was found under partial clustering to be associated with efferocytosis, of which aged microglia had downregulated efferocytosis-associated genes. The impact of aging on microglial efferocytosis was further verified in vitro by doxorubicin (DOX)-induced senescent BV2 microglia, and in vivo by a retinal ischemia/reperfusion (I/R) injury mouse model. In vitro, DOX-treated BV2 microglia had significantly lowered efferocytosis, as well as efferocytosis-related MerTK and Axl protein expression; this was also present in vivo in aged retinas post-I/R injury, with increased co-localization of ionized calcium-binding adapter molecule 1+ microglia with apoptotic retinal cells, along with reduced efferocytosis-related protein expression. Overall, microglial efferocytosis of apoptotic cells decreased with aging, suggesting that modulating this process could serve as a possible therapeutic target for age-related retinal diseases.
Keywords: aging; apoptosis; efferocytosis; microglia; retina; single‐cell RNA sequencing.
© 2025 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
LXR-β regulates microglial efferocytosis and neuroinflammation in CPSP via STAT6 activation.Brain Behav Immun. 2025 Aug 21:106089. doi: 10.1016/j.bbi.2025.106089. Online ahead of print. Brain Behav Immun. 2025. PMID: 40848997
-
Apoptosis recognition receptors regulate skin tissue repair in mice.Elife. 2023 Dec 21;12:e86269. doi: 10.7554/eLife.86269. Elife. 2023. PMID: 38127424 Free PMC article.
-
Single-cell RNA sequencing integrated with bulk RNA sequencing analysis reveals the protective effects of lactate-mediated lactylation of microglia-related proteins on spinal cord injury.CNS Neurosci Ther. 2024 Sep;30(9):e70028. doi: 10.1111/cns.70028. CNS Neurosci Ther. 2024. PMID: 39218784 Free PMC article.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article.
References
-
- Burstyn‐Cohen, T. , and Fresia R.. 2023. “TAM Receptors in Phagocytosis: Beyond the Mere Internalization of Particles.” Immunological Reviews 319, no. 1: 7–26. - PubMed
-
- Chen, J. , Zhang Q., Xu W., et al. 2024. “Baicalein Upregulates Macrophage TREM2 Expression via TrKB‐CREB1 Pathway to Attenuate Acute Inflammatory Injury in Acute‐On‐Chronic Liver Failure.” International Immunopharmacology 139: 112685. - PubMed
MeSH terms
Grants and funding
- 81500713/National Natural Science Foundation of China
- 81970799/National Natural Science Foundation of China
- PL2024H109/Natural Science Foundation of Heilongjiang Province of China
- 20240707020161/Medical and Health Research Projects of Heilongjiang Province
- YQJH2024141/Program for Young Talents of Basic Research in Universities of Heilongjiang Province
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

