Impact of hepatitis C virus genotype on the efficacy of the direct-acting antivirals in chronic kidney disease patients in West Bengal, India
- PMID: 40380327
- PMCID: PMC12083046
- DOI: 10.1186/s12879-025-10947-x
Impact of hepatitis C virus genotype on the efficacy of the direct-acting antivirals in chronic kidney disease patients in West Bengal, India
Abstract
Background: Hepatitis C virus (HCV) infection increases the risk of mortality and morbidity among chronic kidney disease (CKD) patients. However, the advancement of HCV treatment has made this viral infection curable. Thus, the main objective of this study was to comprehend the HCV genotype (GT) distribution and the efficacy of direct-acting antivirals (DAAs) among CKD patients in West Bengal.
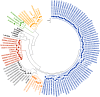
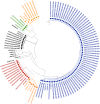
Methods: Over five years (January 2017 to December 2021), 310 HCV sero-reactive patients were enrolled in this observational prospective study. HCV RNA was quantified using qRT-PCR. The partial amplification of the core (405 bp) and NS5B (389 bp) region was performed by nested RT-PCR followed by Sanger sequencing for HCV genotype analysis using the NCBI genotyping tool. The phylogenetic tree was constructed using the MEGA-X tool.
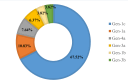
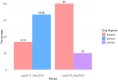
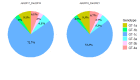
Results: The occurrence of HCV RNA positivity was 50.64% (n = 157), and of these 157 patients, 141 (89.81%) completed the DAAs treatment. The most important observation of the study was the prevalence of uncommon HCV genotype GT-1c (67.52%) followed by 1a, 4a, 3a, 1b, and 3b among CKD patients. The overall DAAs efficacy between January 2017 and December 2018 was ~ 97%, and in January 2019 and December 2021, ~ 95% among CKD patients. At the same time, in these two phases, DAAs efficacy among GT-1c-infected CKD patients was ˜ 96% and ˜ 93%, respectively.
Conclusions: The prevalence of GT-1c among CKD patients was unusual in this geographic region. The overall efficacy of DAAs among the CKD population was encouraging. However, the downtrend of the DAAs efficacy in GT-1c may increase concern among this high-risk group in the future.
Clinical trial: Not applicable.
Keywords: Chronic kidney disease (CKD); Direct-acting antivirals (DAAs); HCV genotype (GT); Hepatitis C virus (HCV).
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent from the participant: The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Ethics Committee of the Indian Council of Medical Research-National Institute of Cholera and Enteric Diseases (ICMR-NICED; recently the name changed to ICMR-NIRBI), Kolkata (Approval number: A-1/2016-IEC; date of approval: 03.10.2016). Informed consent was obtained from all subjects involved in this study. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Efficacy of Different Combinations of Direct-Acting Antivirals Against Different Hepatitis C Virus-Infected Population Groups: An Experience in Tertiary Care Hospitals in West Bengal, India.Viruses. 2025 Feb 16;17(2):269. doi: 10.3390/v17020269. Viruses. 2025. PMID: 40007024 Free PMC article.
-
Evolution of eGFR in chronic HCV patients receiving sofosbuvir-based or sofosbuvir-free direct-acting antivirals.J Hepatol. 2020 May;72(5):839-846. doi: 10.1016/j.jhep.2019.11.014. Epub 2019 Nov 29. J Hepatol. 2020. PMID: 31790766
-
Generic sofosbuvir-based direct-acting antivirals in hepatitis C virus-infected patients with chronic kidney disease.Liver Int. 2018 Dec;38(12):2137-2148. doi: 10.1111/liv.13863. Epub 2018 May 14. Liver Int. 2018. PMID: 29676846
-
Direct-acting antivirals for hepatitis C virus in patients on maintenance dialysis.Int J Artif Organs. 2017 Oct 13;40(10):531-541. doi: 10.5301/ijao.5000613. Epub 2017 Jul 8. Int J Artif Organs. 2017. PMID: 28708211 Review.
-
Hepatitis C and Treatment in Patients with Chronic Kidney Disease.Dis Mon. 2021 Feb;67(2):101017. doi: 10.1016/j.disamonth.2020.101017. Epub 2020 Jun 16. Dis Mon. 2021. PMID: 32553421 Review.
References
-
- Webster AC, Nagler EV, Morton RL, Masson P. Chronic Kidney Disease Lancet. 2017;389(10075):1238–52. 10.1016/S0140-6736(16)32064-5. - PubMed
-
- Yamagata K, Ishida K, Sairenchi T, Takahashi H, Ohba S, Shiigai T, et al. Risk factors for chronic kidney disease in a community-based population: A 10-year follow-up study. Kidney Int. 2007;71(2):159–66. 10.1038/sj.ki.5002017. - PubMed
-
- Francis A, Harhay MN, Ong ACM, Tummalapalli SL, Ortiz A, Fogo AB, et al. Chronic kidney disease and the global public health agenda: an international consensus. Nat Rev Nephrol. 2024;20(July):473–85. 10.1038/s41581-024-00820-6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

