Deep transfer learning approach for automated cell death classification reveals novel ferroptosis-inducing agents in subsets of B-ALL
- PMID: 40382332
- PMCID: PMC12085637
- DOI: 10.1038/s41419-025-07704-y
Deep transfer learning approach for automated cell death classification reveals novel ferroptosis-inducing agents in subsets of B-ALL
Abstract
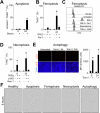
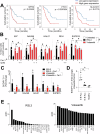
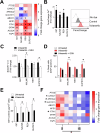
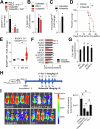
Ferroptosis is a recently described type of regulated necrotic cell death whose induction has anti-cancer therapeutic potential, especially in hematological malignancies. However, efforts to uncover novel ferroptosis-inducing therapeutics have been largely unsuccessful. In the current investigation, we classified brightfield microscopy images of tumor cells undergoing defined modes of cell death using deep transfer learning (DTL). The trained DTL network was subsequently combined with high-throughput pharmacological screening approaches using automated live cell imaging to identify novel ferroptosis-inducing functions of the polo-like kinase inhibitor volasertib. Secondary validation showed that subsets of B-cell acute lymphoblastic leukemia (B-ALL) cell lines, namely 697, NALM6, HAL01, REH and primary patient B-ALL samples were sensitive to ferroptosis induction by volasertib. This was accompanied by an upregulation of ferroptosis-related genes post-volasertib treatment in cell lines and patient samples. Importantly, using several leukemia models, we determined that volasertib delayed tumor growth and induced ferroptosis in vivo. Taken together, we have applied DTL to automated live-cell imaging in pharmacological screening to identify novel ferroptosis-inducing functions of a clinically relevant anti-cancer therapeutic.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: Experiments were performed under the authorization of LANUV in accordance with German law for animal protection. Patient consent for publication: This study involves human participants after obtaining informed consent in accordance with the Declaration of Helsinki. The experiments were approved by the ethics committee of the Heinrich Heine University medical faculty (Study Nr.: 2019-566).
Figures


References
MeSH terms
Substances
Grants and funding
- DJCLS 18R/2021/José Carreras Leukämie-Stiftung (Deutsche José Carreras Leukämie-Stiftung)
- DJCLS 18R/2021/José Carreras Leukämie-Stiftung (Deutsche José Carreras Leukämie-Stiftung)
- DJCLS 07/19/José Carreras Leukämie-Stiftung (Deutsche José Carreras Leukämie-Stiftung)
- 01KD2410A (EDI-4-ALL)/Bundesministerium für Bildung und Forschung (Federal Ministry of Education and Research)
- 01KD2410A (EDI-4-ALL)/Bundesministerium für Bildung und Forschung (Federal Ministry of Education and Research)
- A07/University of Cologne | Center for Molecular Medicine Cologne, University of Cologne (CMMC, UoC)
- CRC1530, project ID 455784452/Deutsche Forschungsgemeinschaft (German Research Foundation)
- CRC1403, project ID 414786233/Deutsche Forschungsgemeinschaft (German Research Foundation)
- CRC1310, project ID 325931972/Deutsche Forschungsgemeinschaft (German Research Foundation)
- CRC1399, project ID 413326622/Deutsche Forschungsgemeinschaft (German Research Foundation)
- SPP2306, project ID 461704389/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 495318549/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 270650915/Deutsche Forschungsgemeinschaft (German Research Foundation)
- 528968169/Deutsche Forschungsgemeinschaft (German Research Foundation)
- Priority Program Cancer Prevention/Deutsche Krebshilfe (German Cancer Aid)
LinkOut - more resources
Full Text Sources

