MR Spectroscopic Imaging of Hyperpolarized 129-Xenon in the Dissolved-Phase to Determine Regional Chemical Shifts of Hyperoxia in Healthy Porcine Lungs
- PMID: 40384034
- PMCID: PMC12086352
- DOI: 10.1002/nbm.70063
MR Spectroscopic Imaging of Hyperpolarized 129-Xenon in the Dissolved-Phase to Determine Regional Chemical Shifts of Hyperoxia in Healthy Porcine Lungs
Abstract
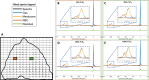
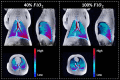
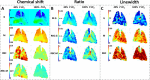
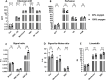
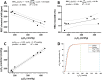
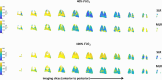
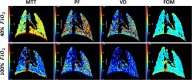
Lung MRI with hyperpolarized xenon (129Xe) gas reveals key characteristics of pulmonary physiology such as ventilation and alveolar-capillary gas transfer. Magnetic resonance spectroscopic imaging (MRSI) offers insights into regional oxygenation saturation (sO2) through chemical shift changes related to xenon-hemoglobin binding. The similarity between porcine and human anatomy and physiology, particularly in terms of lung volume, airway structure, and alveolar-capillary microstructure, offers the opportunity to investigate physiological effects linked to oxygen supply using 129Xe MRSI. We hypothesize that 129Xe MRSI can detect regional chemical shift changes related to red blood cell oxygenation and arterial oxygen partial pressure (paO2) in a porcine model. Imaging was performed on a 3-T clinical MRI scanner on four healthy pigs mechanically ventilated at fractional inspired oxygen levels (FiO2) of 40% and 100%. Dissolved-phase images were acquired using a 3D Cartesian MRSI sequence with a spherical sampling pattern in a matrix size of 28 × 28 × 6. A spectrally tailored RF pulse excited the dissolved and gaseous phases with flip angles of 10° and 0.1°, respectively. Repetition time was 7.4 ms resulting in a total acquisition time of 18 s. In addition, 129Xe ventilation, pulmonary anatomical scans, dynamic contrast-enhanced perfusion, and arterial blood gas were measured at each FiO2. Pair-wise comparisons were performed between inspired oxygen levels, along with linear regression analysis of paO2 and dissolved-phase chemical shift imaging. Porcine lung lobes were segmented, and two-way ANOVA were performed to evaluate regional effects of oxygen concentrations. Arterial blood gas and cardiopulmonary measures showed an increase in paO2 with the increase in FiO2. Ventilation defect percentage and perfusion metrics did not significantly change with higher oxygen concentration. Dissolved-phase ratios of red blood cells (RBC) to membrane increased with higher oxygen concentration. Increasing inspired oxygen resulted in a lower RBC chemical shift and increased linewidth, indicating RBC measures are sensitive to paO2. Simple linear regression analysis of RBC chemical shift and a multiple linear regression model including linewidth were applied for regional paO2 maps. Regional effects of oxygen were confirmed in the segmented lung lobes. Dissolved-phase 129Xe chemical shift of RBC decreased linearly with paO2 in healthy porcine lungs. Regional chemical shift, linewidth, and signal ratio changes were determined in dissolved-phase imaging of RBC at 40% and 100% FiO2. Our data suggest that regional paO2 prediction is possible with a multiple linear regression model including RBC chemical shift and linewidth as combined effect of oxygen across animal lung lobes affects regions differently.
Keywords: arterial oxygen partial pressure; blood oxygenation saturation; chemical shift imaging; dissolved‐phase imaging; hyperpolarized xenon gas; magnetic resonance imaging; porcine animal model.
© 2025 The Author(s). NMR in Biomedicine published by John Wiley & Sons Ltd.
Conflict of interest statement
M.V. and R.F.S. are employees of GE HealthCare. The authors report no conflicts of interest.
Figures




Similar articles
-
Imaging gas-exchange lung function and brain tissue uptake of hyperpolarized 129 Xe using sampling density-weighted MRSI.Magn Reson Med. 2023 Jun;89(6):2217-2226. doi: 10.1002/mrm.29602. Epub 2023 Feb 6. Magn Reson Med. 2023. PMID: 36744585
-
Dissolved 129 Xe lung MRI with four-echo 3D radial spectroscopic imaging: Quantification of regional gas transfer in idiopathic pulmonary fibrosis.Magn Reson Med. 2021 May;85(5):2622-2633. doi: 10.1002/mrm.28609. Epub 2020 Nov 30. Magn Reson Med. 2021. PMID: 33252157
-
Removal of hyperpolarized 129 Xe gas-phase contamination in spectroscopic imaging of the lungs.Magn Reson Med. 2018 Dec;80(6):2586-2597. doi: 10.1002/mrm.27349. Epub 2018 Jun 12. Magn Reson Med. 2018. PMID: 29893992 Free PMC article.
-
Hyperpolarized 129Xe MRI and Spectroscopy: Quantitative Measurements, Results, and Emerging Opportunities.Radiol Cardiothorac Imaging. 2025 Aug;7(4):e240562. doi: 10.1148/ryct.240562. Radiol Cardiothorac Imaging. 2025. PMID: 40772846 Review.
-
In vivo methods and applications of xenon-129 magnetic resonance.Prog Nucl Magn Reson Spectrosc. 2021 Feb;122:42-62. doi: 10.1016/j.pnmrs.2020.11.002. Epub 2020 Dec 9. Prog Nucl Magn Reson Spectrosc. 2021. PMID: 33632417 Free PMC article. Review.
References
-
- Niedbalski P. J., Hall C. S., Castro M., et al., “Protocols for Multi‐Site Trials Using Hyperpolarized 129Xe MRI for Imaging of Ventilation, Alveolar‐Airspace Size, and Gas Exchange: A Position Paper From the 129Xe MRI Clinical Trials Consortium,” Magnetic Resonance in Medicine 86, no. 6 (2021): 2966–2986, 10.1002/mrm.28985. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

