Cenobamate in Real-Word Scenario: Results on Efficacy, Side Effects, and Retention Rate in a Single Center Retrospective Study
- PMID: 40384045
- PMCID: PMC12086305
- DOI: 10.1002/brb3.70567
Cenobamate in Real-Word Scenario: Results on Efficacy, Side Effects, and Retention Rate in a Single Center Retrospective Study
Abstract
Background: Pharmacoresistance imposes a high burden on people with epilepsy (PWE). Recently authorized cenobamate (CNB) offers new hope with high efficacy reported in phase III and early real-world studies. Here, we present data from a reasonably sized monocentric cohort, complementing the knowledge derived from clinical practice.
Methods: We retrospectively analyzed medical records of all PWE treated with CNB from market entry to July 31, 2023.
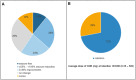
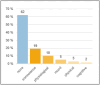
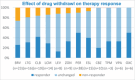
Results: After an average of 1.1 years, follow-up data were available for 262 out of 280 PWE, who received at least one dose of CNB. The average CNB dose was 183 ± 98 mg/d, with a mean number of anti-seizure medications (ASM) of 2.9 ± 1 per patient. A total of 36% of the patients showed ≥ 50% reduction in seizure frequency (10.7% were seizure-free), whilst 12.3% reported increased seizure frequency. Seizure freedom was associated with concomitant perampanel or GABA receptor modulators. No predictors of treatment response were found. Side effects were reported by 38%, alertness issues being most prevalent (19%). The retention rate amounted to 72% and was associated with response status, dose of CNB, side effects, and age at CNB introduction. Among those who discontinued CNB, 55% experienced side effects and 89% showed no meaningful seizure reduction. Regarding co-medication, the withdrawal of lamotrigine, brivaracetam, clobazam, or lacosamide was associated with higher rates of non-response.
Conclusions: In this large cohort of 262 PWE, CNB proved very efficient with a high retention rate over one year. Co-medication with perampanel or GABA receptor modulators was linked to seizure-freedom. The overall positive impression of CNB is further supported.
Keywords: cenobamate; efficacy; real‐world data; retention rate; side effects.
© 2025 The Author(s). Brain and Behavior published by Wiley Periodicals LLC.
Figures
References
-
- Carreño, M. , Gil‐Nagel A., Serratosa J. M., Toledo M., Rodriguez‐Uranga J. J., and Villanueva V. 2024. “Spanish Consensus on the Management of Concomitant Antiseizure Medications When Using Cenobamate in Adults With Drug‐resistant Focal Seizures.” Epilepsia Open 9, no. 3: 1051–1058. 10.1002/epi4.12936. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical