Advancements in AI-driven drug sensitivity testing research
- PMID: 40384974
- PMCID: PMC12081381
- DOI: 10.3389/fcimb.2025.1560569
Advancements in AI-driven drug sensitivity testing research
Abstract
Antimicrobial resistance (AMR) constitutes a significant global public health challenge, posing a serious threat to human health. In clinical practice, physicians frequently resort to empirical antibiotic therapy without timely Antimicrobial Susceptibility Testing (AST) results. This practice, however, may induce resistance mutations in pathogens due to genetic pressure, thereby complicating infection control efforts. Consequently, the rapid and accurate acquisition of AST results has become crucial for precision treatment. In recent years, advancements in medical testing technology have led to continuous improvements in AST methodologies. Concurrently, emerging artificial intelligence (AI) technologies, particularly Machine Learning(ML) and Deep Learning(DL), have introduced novel auxiliary diagnostic tools for AST. These technologies can extract in-depth information from imaging and laboratory data, enabling the swift prediction of pathogen antibiotic resistance and providing reliable evidence for the judicious selection of antibiotics. This article provides a comprehensive overview of the advancements in research concerning pathogen AST and resistance detection methodologies, emphasizing the prospective application of artificial intelligence and machine learning in predicting drug sensitivity tests and pathogen resistance. Furthermore, we anticipate future directions in AST prediction aimed at reducing antibiotic misuse, enhancing treatment outcomes for infected patients, and contributing to the resolution of the global AMR crisis.
Keywords: antimicrobial resistance; antimicrobial susceptibility testing; artificial intelligence; machine learning; whole genome sequencing.
Copyright © 2025 Liao, Xie, Zhang, Lu and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest
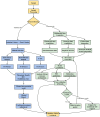
Figures
Similar articles
-
Artificial intelligence in bacterial diagnostics and antimicrobial susceptibility testing: Current advances and future prospects.Biosens Bioelectron. 2025 Jul 15;280:117399. doi: 10.1016/j.bios.2025.117399. Epub 2025 Mar 19. Biosens Bioelectron. 2025. PMID: 40184880 Review.
-
Artificial intelligence in predicting pathogenic microorganisms' antimicrobial resistance: challenges, progress, and prospects.Front Cell Infect Microbiol. 2024 Nov 1;14:1482186. doi: 10.3389/fcimb.2024.1482186. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39554812 Free PMC article. Review.
-
From Data to Decisions: Leveraging Artificial Intelligence and Machine Learning in Combating Antimicrobial Resistance - a Comprehensive Review.J Med Syst. 2024 Aug 1;48(1):71. doi: 10.1007/s10916-024-02089-5. J Med Syst. 2024. PMID: 39088151 Free PMC article. Review.
-
Advances in genotypic antimicrobialresistance testing: a comprehensive review.Sci China Life Sci. 2025 Jan;68(1):130-143. doi: 10.1007/s11427-023-2570-4. Epub 2024 Sep 18. Sci China Life Sci. 2025. PMID: 39300049 Review.
-
Disc Diffusion Reader: an AI-powered potential solution to combat antibiotic resistance in developing countries.J Infect Dev Ctries. 2025 May 31;19(5):699-711. doi: 10.3855/jidc.21108. J Infect Dev Ctries. 2025. PMID: 40452528
References
-
- Ambade S. S., Gupta V. K., Bhole R. P., Khedekar P. B., Chikhale R. V. (2023). A review on five and six-membered heterocyclic compounds targeting the penicillin-binding protein 2 (PBP2A) of methicillin-resistant staphylococcus aureus (MRSA). Molecules 28, 7008. doi: 10.3390/molecules28207008 - DOI - PMC - PubMed
-
- Anh Tuan D., Uyen P. V. N., Masak J. (2024). Hybrid quorum sensing and machine learning systems for adaptive synthetic biology: toward autonomous gene regulation and precision therapies. doi: 10.20944/preprints202410.1551.v1 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

