Conducting a registry-based randomised trial (REDOX) in chronic respiratory failure: experiences and advice
- PMID: 40386081
- PMCID: PMC12082728
- DOI: 10.1080/20018525.2025.2502237
Conducting a registry-based randomised trial (REDOX) in chronic respiratory failure: experiences and advice
Abstract
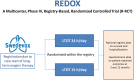
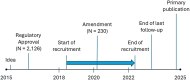
Registry-based randomised controlled trials (R-RCTs) represent a paradigm shift in research, with the potential to accomplish pragmatic but large trials with high external validity. In this paper, we review our experiences from planning and performing the REgistry-based randomised controlled trial of treatment Duration and mortality in long-term OXygen therapy (REDOX) trial, the first R-RCT within respiratory medicine. The REDOX study compared the two established treatment options of home oxygen 15 and 24 h per day. Previous recommendations to use oxygen for at least 15 h but preferably 24 h per day were based on a non-randomised comparison of two different studies. We hypothesised that oxygen 24 h/day was non-superior to 15 h/day and used the Swedish National Registry for Respiratory Failure (Swedevox) to perform an R-RCT showing that home oxygen 24 h/day does not improve survival, hospitalisation or patient-reported outcomes within 1 year. We describe the entire procedure of REDOX from planning to publication and use it to discuss challenges and potential solutions for future R-RCTs. In summary, common features of R-RCTs are the use of a registry for identification and randomisation of participants and for reporting and collecting baseline and outcome data, and the design is typically used to compare two treatment options. Important strengths are high generalisability, low cost, feasibility for consecutive recruitment in clinical practice, and high completeness of follow-up. Limitations include that coverage, completeness and accuracy of baseline data may differ between registries. Specific challenges (and solutions) in REDOX were addressing an important question (pragmatic clinical trials), management (clinical research support teams), costs (using registry-based infrastructure), different electronic data capture systems (posttrial linkage), slow recruitment (amendment of protocol) and resistance to challenge treatment traditions.
Keywords: hospitalisation; hypoxemia; mortality; oxygen therapy; trial design.
© 2025 The Author(s). Published by Informa UK Limited, trading as Taylor & Francis Group.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures
Similar articles
-
REgistry-based randomized controlled trial of treatment and Duration and mortality in long-term OXygen therapy (REDOX) study protocol.BMC Pulm Med. 2019 Feb 26;19(1):50. doi: 10.1186/s12890-019-0809-7. BMC Pulm Med. 2019. PMID: 30808321 Free PMC article.
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
A Phase I/II Clinical Trial to evaluate the efficacy of baricitinib to prevent respiratory insufficiency progression in onco-hematological patients affected with COVID19: A structured summary of a study protocol for a randomised controlled trial.Trials. 2021 Feb 5;22(1):116. doi: 10.1186/s13063-021-05072-4. Trials. 2021. PMID: 33546739 Free PMC article.
-
Electronic self-reporting of adverse events for patients undergoing cancer treatment: the eRAPID research programme including two RCTs.Southampton (UK): NIHR Journals Library; 2022 Feb. Southampton (UK): NIHR Journals Library; 2022 Feb. PMID: 35138783 Free Books & Documents. Review.
-
Sutureless Aortic Valve Replacement for Treatment of Severe Aortic Stenosis: A Single Technology Assessment of Perceval Sutureless Aortic Valve [Internet].Oslo, Norway: Knowledge Centre for the Health Services at The Norwegian Institute of Public Health (NIPH); 2017 Aug 25. Report from the Norwegian Institute of Public Health No. 2017-01. Oslo, Norway: Knowledge Centre for the Health Services at The Norwegian Institute of Public Health (NIPH); 2017 Aug 25. Report from the Norwegian Institute of Public Health No. 2017-01. PMID: 29553663 Free Books & Documents. Review.
References
Publication types
LinkOut - more resources
Full Text Sources