Mucinous breast cancer organoids: an in vitro research model
- PMID: 40388071
- PMCID: PMC12089555
- DOI: 10.1007/s12672-025-02586-0
Mucinous breast cancer organoids: an in vitro research model
Abstract
Background: Pure mucinous breast cancer is an uncommon form of cancer characterized by a low metastatic rate and a generally favorable prognosis. However, some patients may experience lymph node metastasis, leading to a worse prognosis. Currently, there is no reliable in vitro model available to effectively address the heterogeneity of pure mucinous breast cancer.
Methods: We obtained surgical tumor samples from a 64-year-old Chinese female patient diagnosed with pure mucinous breast cancer to establish patient-derived organoids. Using these organoids, we performed histological staining, drug testing and single-cell RNA-Seq analysis.
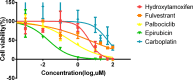
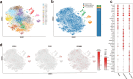
Results: We accomplished the establishment of a patient-derived mucinous breast cancer organoid model from a Chinese female. Hematoxylin and eosin staining, along with immunohistochemistry, revealed histology and protein expression (ER, PR, HER2 and Ki-67) at early passages similar to the original breast cancer tissue. Single-cell RNA sequencing at passage 7 identified 17 cell clusters, which were assigned to three cell types based on marker genes. This showed that most ER-positive luminal cells had been replaced by ER-negative basal-like cells at passage 7. We tested drug sensitivity to five antitumor drugs at passage 5. The organoids showed the highest sensitivity to Epirubicin and the lowest sensitivity to Carboplatin.
Conclusions: This is the first reported case of a mucinous breast cancer organoid. Our experimental results indicate that this model exhibits similar characteristics to the original tissue at early passages. Organoids at early passages could be a promising tool for clinical drug screening and further scientific research.
Keywords: Heterogeneity; Mucinous breast cancer; Organoid; Single-cell RNA-Seq analysis.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Approval was obtained from the Ethics Committee of The Second Hospital of Dalian Medical University. The methods employed in this research comply with the principles of the Declaration of Helsinki. Informed consent was secured from each participant involved in the study. Consent for publication: Patient signed informed consent regarding publishing her data and photographs. Competing interests: The authors declare no competing interests.
Figures


References
Grants and funding
- 82203800/National Natural Science Foundation of China
- 82072934/National Natural Science Foundation of China
- 2023-MSLH-022/Natural Science Foundation of Liaoning Province
- DYLX22014/Educational Reform Project of Dalian Medical University
- 2022LCYJYB03/1+X program of the Second Hospital of Dalian Medical University
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
