Efficacy and Safety of Eptinezumab in Episodic Cluster Headache: A Randomized Clinical Trial
- PMID: 40388178
- PMCID: PMC12090066
- DOI: 10.1001/jamaneurol.2025.1317
Efficacy and Safety of Eptinezumab in Episodic Cluster Headache: A Randomized Clinical Trial
Abstract
Importance: Cluster headache, characterized by bouts of excruciating pain attacks, detrimentally affects health and quality of life. Eptinezumab is an anticalcitonin gene-related peptide monoclonal antibody approved for migraine prevention.
Objective: To evaluate the efficacy and safety of eptinezumab in the preventive treatment of episodic cluster headache.
Design, setting, and participants: This double-blind, placebo-controlled, randomized (1:1) clinical trial (Eptinezumab in Participants With Episodic Cluster Headache [ALLEVIATE]) was conducted between December 2020 and October 2023. Results are from the initial 4-week randomized phase. The study took place at 64 sites across Europe, the US, and Japan. Included were adults (aged 18-75 years) with a history of episodic cluster headache for 1 or more years (with bouts lasting ≥6 weeks when untreated) and previous acute and preventive medication use.
Interventions: Eptinezumab, 400 mg, or placebo (intravenous infusion).
Main outcomes and measures: The primary end point was the change from baseline in the number of weekly attacks in weeks 1 to 2. Safety was assessed using treatment-emergent adverse events.
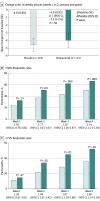
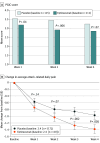
Results: Of 628 total participants screened, 320 entered the second screening period, and 231 met eligibility criteria. Of the 231 participants randomized (eptinezumab, n = 118; placebo, n = 113), 215 (93%) completed the placebo-controlled period. The participant mean (SD) age was 44 (11) years, and 178 of 229 were male (78%). At baseline, the mean (SD) weekly attacks were 15.2 (8.1) in the eptinezumab group and 15.7 (8.3) in the placebo group. There was no statistically significant difference between eptinezumab and placebo in the change from baseline in the number of weekly attacks over weeks 1 to 2 (least-squares mean [SE], -4.0 [0.93] vs -4.6 [0.89]; between-group difference, 0.7; 95% CI, -1.3 to 2.6; P = .50). More eptinezumab-treated participants achieved 50% or greater response vs placebo over week 2 (50.9% [54 of 106] vs 37.3% [41 of 110]; odds ratio [OR], 1.77; 95% CI, 1.03-3.07; P =.04), week 3 (62.5% [65 of 104] vs 43.8% [49 of 112]; OR, 2.26; 95% CI, 1.30-3.97; P =.004), and week 4 (66.7% [68 of 102] vs 50.5% [54 of 107]; OR, 2.14; 95% CI, 1.21-3.83; P =.009). Eptinezumab showed numerically larger improvements than placebo for 75% or greater response, average daily pain scores, and across other patient-reported outcomes. Treatment-emergent adverse events occurred in 25.0% of patients (28 of 112) receiving eptinezumab and 26.5% of patients (31 of 117) receiving placebo.
Conclusions and relevance: Among adults with episodic cluster headache, eptinezumab did not significantly reduce the number of attacks vs placebo, although it was associated with numerically higher responder rates and improvements in average daily pain and patient-reported outcomes. Eptinezumab was generally well tolerated.
Trial registration: ClinicalTrials.gov Identifier: NCT04688775.
Conflict of interest statement
Figures
References
-
- Freeman E, Adair M, Beeler D, et al. Patient-identified burden and unmet needs in patients with cluster headache: An evidence-based qualitative literature review. Cephalalgia Rep. 2022;5:1-17. doi: 10.1177/25158163221096866 - DOI
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

