Wnt/β-catenin activation by mutually exclusive FBXW11 and CTNNB1 hotspot mutations drives salivary basal cell adenoma
- PMID: 40389436
- PMCID: PMC12089348
- DOI: 10.1038/s41467-025-59871-3
Wnt/β-catenin activation by mutually exclusive FBXW11 and CTNNB1 hotspot mutations drives salivary basal cell adenoma
Abstract
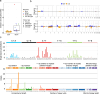
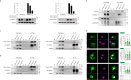
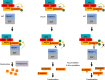
Basal cell adenoma (BCA) and basal cell adenocarcinoma (BCAC) of the salivary gland are rare tumours that can be difficult to distinguish from each other and other salivary gland tumour subtypes. Using next-generation sequencing, we identify a recurrent FBXW11 missense mutation (p.F517S) in BCA that is mutually exclusive with the previously reported CTNNB1 p.I35T gain-of-function (GoF) mutation with these mutations collectively accounting for 94% of BCAs. In vitro, mutant FBXW11 is characterised by defective binding to β-catenin and higher protein levels within the nucleus. This is consistent with the increased nuclear expression of β-catenin and activation of the Wnt/β-catenin pathway. The genomic profiles of BCAC are distinct from BCA, with hotspot DICER1 and HRAS mutations and putative driver mutations affecting PI3K/AKT and NF-κB signalling pathway genes. These findings have important implications for the diagnosis and treatment of BCA and BCAC, which, despite histopathologic overlap, may be unrelated entities.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors have no conflicts to declare.
Figures




Similar articles
-
Wnt/β-catenin signal alteration and its diagnostic utility in basal cell adenoma and histologically similar tumors of the salivary gland.Pathol Res Pract. 2018 Apr;214(4):586-592. doi: 10.1016/j.prp.2017.12.016. Epub 2018 Jan 3. Pathol Res Pract. 2018. PMID: 29496310
-
CTNNB1 mutations in basal cell adenoma of the salivary gland.J Formos Med Assoc. 2018 Oct;117(10):894-901. doi: 10.1016/j.jfma.2017.11.011. Epub 2017 Dec 7. J Formos Med Assoc. 2018. PMID: 29224720
-
Correlation between basal cell adenoma and basal cell adenocarcinoma of the salivary gland: a histomorphological and molecular review of 129 cases.Virchows Arch. 2025 Jul;487(1):75-86. doi: 10.1007/s00428-025-04120-7. Epub 2025 May 13. Virchows Arch. 2025. PMID: 40360858 Free PMC article.
-
Infarction of basal cell adenoma of the parotid gland: First case report and literature review on CTNNB1 I35T mutations in salivary basal cell neoplasms.Oral Oncol. 2023 Oct;145:106534. doi: 10.1016/j.oraloncology.2023.106534. Epub 2023 Jul 28. Oral Oncol. 2023. PMID: 37517215 Review.
-
Basal Cell Adenoma and Basal Cell Adenocarcinoma.Surg Pathol Clin. 2021 Mar;14(1):25-42. doi: 10.1016/j.path.2020.09.005. Epub 2021 Jan 5. Surg Pathol Clin. 2021. PMID: 33526221 Review.
References
-
- Robinson, R. A. Basal cell adenoma and basal cell adenocarcinoma. Surg. Pathol. Clin.14, 25–42 (2021). - PubMed
-
- WHO Classification of Tumours Editorial Board. Head and Neck Tumours (WHO Classification of Tumours Series, 5th Ed). 9 (International Agency for Research on Cancer, Lyon (France), 2022).
-
- Scott, A. R., Faquin, W. C. & Deschler, D. G. Parotid mass in a woman with multiple cutaneous cylindromas. Head. Neck32, 684–687 (2010). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

