Targeting Lp-PLA2 inhibits profibrotic monocyte-derived macrophages in silicosis through restoring cardiolipin-mediated mitophagy
- PMID: 40389600
- PMCID: PMC12206922
- DOI: 10.1038/s41423-025-01288-5
Targeting Lp-PLA2 inhibits profibrotic monocyte-derived macrophages in silicosis through restoring cardiolipin-mediated mitophagy
Abstract
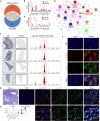
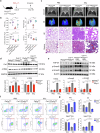
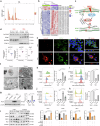
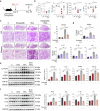
Monocyte-derived macrophages (MoMacs) are the most important effector cells that cause pulmonary fibrosis. However, the characteristics of MoMac differentiation in silicosis and the mechanisms by which MoMacs affect the progression of pulmonary fibrosis remain unclear. Integration of single-cell and spatial transcriptomic analyses revealed that the silicosis niche was occupied by a subset of MoMacs, identified as Spp1hiMacs, which remain in an immature transitional state of differentiation during silicosis. This study investigated the mechanistic foundations of mitochondrial damage induced by the lipoprotein-associated phospholipase A2 (Lp-PLA2, encoded by Pla2g7)-acyl-CoA:lysocardiolipin acyltransferase-1 (ALCAT1)-cardiolipin (CL) signaling pathway, which interferes with Spp1hiMac differentiation. We demonstrated that in SiO2-induced MoMacs, Lp-PLA2 induces abnormal CL acylation through the activation of ALCAT1, resulting in impaired mitochondrial localization of PINK1 and LC3B and mitochondrial autophagy defects. Simultaneously, lysosomal dysfunction causes the release of the lysosomal protein cathepsin B into the cytoplasm, which involves M1 and M2 macrophage polarization and the activation of proinflammatory and profibrotic pathways. Furthermore, we assessed the efficacy of the Lp-PLA2 inhibitor darapladib in ameliorating silica-induced pulmonary fibrosis in a murine model. Our findings enhance our understanding of silicosis pathogenesis and offer promising opportunities for developing targeted therapies to mitigate fibrotic progression and maintain lung function in affected individuals.
Keywords: Cardiolipin; Lipoprotein-associated phospholipase A2; Mitophagy; Monocyte-derived macrophages; Pulmonary fibrosis.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures




Similar articles
-
Repeated Silica exposures lead to Silicosis severity via PINK1/PARKIN mediated mitochondrial dysfunction in mice model.Cell Signal. 2024 Sep;121:111272. doi: 10.1016/j.cellsig.2024.111272. Epub 2024 Jun 27. Cell Signal. 2024. PMID: 38944258
-
Degradation of oxidized phospholipids by lysosomal phospholipase A2 regulates pulmonary fibrosis.Am J Physiol Lung Cell Mol Physiol. 2025 Aug 1;329(2):L214-L223. doi: 10.1152/ajplung.00038.2025. Epub 2025 Jun 26. Am J Physiol Lung Cell Mol Physiol. 2025. PMID: 40569270 Free PMC article.
-
Erchen decoction alleviates silicosis by attenuating ferroptosis and fibrosis in alveolar macrophages via modulating the P53/HMOX1 pathway.J Ethnopharmacol. 2025 Aug 29;352:120227. doi: 10.1016/j.jep.2025.120227. Epub 2025 Jul 4. J Ethnopharmacol. 2025. PMID: 40617365
-
Genetic invalidation of Lp-PLA2 as a therapeutic target: Large-scale study of five functional Lp-PLA2-lowering alleles.Eur J Prev Cardiol. 2017 Mar;24(5):492-504. doi: 10.1177/2047487316682186. Epub 2016 Dec 8. Eur J Prev Cardiol. 2017. PMID: 27940953 Free PMC article.
-
Cardiolipin Remodeling in Cardiovascular Diseases: Implication for Mitochondrial Dysfunction.Acta Physiol (Oxf). 2025 Jul;241(7):e70073. doi: 10.1111/apha.70073. Acta Physiol (Oxf). 2025. PMID: 40530586 Review.
References
-
- Hoy RF, Chambers DC. Silica-related diseases in the modern world. Allergy. 2020;75:2805–17. - PubMed
-
- Elderbrook M, Harrison R, Grajewski B, Tomasallo C, Meiman J. Silicosis: Emerging Trends and How to Screen for Early Detection. WMJ. 2023;122:114–7. - PubMed
-
- Zhu Y, Duan XY, Cheng YQ, Yao XJ, Xu H, Zhang KS, et al. Evaluation of differential serum expression of three factors and pulmonary function in patients with silicosis. Int J Occup Med Environ Health. 2021;34:527–40. - PubMed
MeSH terms
Substances
Grants and funding
- No. 82370072/National Natural Science Foundation of China (National Science Foundation of China)
- No. 81870056/National Natural Science Foundation of China (National Science Foundation of China)
- No. 9206810167/National Natural Science Foundation of China (National Science Foundation of China)
- No. 82003406/National Natural Science Foundation of China (National Science Foundation of China)
- 2023M733985/China Postdoctoral Science Foundation
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

