S100B Mitigates Cytoskeletal and Mitochondrial Alterations in a Glial Cell Model of Autosomal Recessive Spastic Ataxia of Charlevoix-Saguenay
- PMID: 40389788
- PMCID: PMC12367932
- DOI: 10.1007/s12035-025-05057-3
S100B Mitigates Cytoskeletal and Mitochondrial Alterations in a Glial Cell Model of Autosomal Recessive Spastic Ataxia of Charlevoix-Saguenay
Abstract
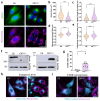
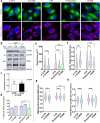
Autosomal Recessive Spastic Ataxia of Charlevoix-Saguenay (ARSACS) is an early-onset neurological disorder caused by mutations in the SACS gene, resulting in the loss of sacsin function. Sacsin is a multidomain protein that plays key roles in chaperone regulation, protein quality control, and neurofilament dynamics. Sacsin deficiency leads to disruption of intermediate filament and mitochondrial networks. S100B, a multifunctional brain-enriched protein, exhibits protective neuroprotective functions that include chaperone activity and interactions with filament proteins and mitochondria. In this study, we used an established astroglial C6 cell model of ARSACS to investigate the potential compensatory effects of S100B on sacsin loss with respect to neurofilament integrity and mitochondrial morphological and functional hallmarks. Our results demonstrate that sacsin deletion induces S100B upregulation at both mRNA and protein levels, with the S100B protein colocalizing with perinuclear nestin aggregates and filamentous mitochondria networks. Genetic silencing and pharmacological inhibition of S100B exacerbate filament protein aggregation and mitochondrial defects, while supplementation with exogenous recombinant S100B improves ARSACS hallmarks, including decreased nestin aggregates. These findings provide evidence for functional compensation of sacsin loss by S100B in glial cells, and suggests a potential role for glial cells in ARSACS.
Keywords: ARSACS; Astroglia; Intermediate filaments; Mitochondria; Molecular chaperones; S100B; Sacsin.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures

References
-
- Salem IH, Blais M, Zuluaga-Sánchez VM et al (2025) ARSACS: clinical features, pathophysiology and iPS-derived models. Cerebellum 24(1):24. 10.1007/s12311-024-01777-9 - PubMed
-
- Engert J, Bérubé P, Mercier J, et al (2000) ARSACS, a spastic ataxia common in northeastern Québec, is caused by mutations in a new gene encoding an 11.5-kb ORF. Nat Genet 24:120–125. 10.1038/72769 - PubMed
-
- Aly KA, Moutaoufik MT, Zilocchi M et al (2022) Insights into SACS pathological attributes in autosomal recessive spastic ataxia of Charlevoix-Saguenay (ARSACS)☆. Curr Opin Chem Biol 71:102211. 10.1016/j.cbpa.2022.102211 - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
- 2024.02710.BD/Fundação para a Ciência e a Tecnologia
- SFRH/BD/133220/2017/Fundação para a Ciência e a Tecnologia
- https://doi.org/10.54499/UIDB/04046/2020/Fundação para a Ciência e a Tecnologia
- https://doi.org/10.54499/UIDP/04046/2020/Fundação para a Ciência e a Tecnologia
- PPBI-POCI-01-0145-FEDER-022122/European Regional Development Fund
LinkOut - more resources
Full Text Sources
Miscellaneous

